BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
URL: http://ijt.arakmu.ac.ir/article-1-798-en.html
Introduction
In recent years, increasing changes in many aspects of dietary habits in humans, especially the high consumption of processed foods loaded with preservatives and additives have raised concerns about the relationship with reproductive system disorders. Sulfite compounds, such as Sodium Metabisulfite (SMB) are used increasingly in food and pharmaceutical industry. The daily consumption of sulfite in foods and beverages by people is more than the acceptable safe level (0-0.7 mg/kg) and may reach 180-200 mg/kg [1]. In spite of the apparent safety of sulfite additives, numerous reports have been published on the adverse effects of sulfite exposure, leading to dermatological, gastrointestinal, respiratory and renal disorders and may cause life-threatening anaphylactic and asthmatic reactions [2-4]. In addition, sulfites generate free radicals and are strong oxidants of biomolecules [5].
It has been suggested that the oxidative stress creates an environment that leads to abnormal physiological reactions in women, causing diseases of reproductive system and complicating pregnancies [6]. In a previous study, our group reported that sulfite ingestion decreased the number of normal ovarian follicles while increasing the degenerate one [7]. Studies in mice and rats have suggested that differential follicle counts are the best markers to determine the extent of ovarian injury induced by xenobiotics [8, 9].
Previous studies have shown that natural antioxidants have great potential for protecting DNA and other biomolecules from damages caused by oxidation [10, 11]. Ginger (Zingiber officinale) with a wide range of biological properties contains high amount of antioxidants, such as gingerol and shogaol [12, 13]. Ginger has been shown to lower lipid peroxidation by maintaining antioxidant enzymes activities and increasing glutathione level in the rat blood [14]. Its beneficial effect has been observed on ovarian folliculogenesis [15], ovarian cancer by inhibiting growth and modulating secretion of angiogenic factors [16], and cisplatin-induced reproductive damages by normalizing antioxidant enzyme activities in rat testes [17].
Due to high consumption of processed and fast foods containing various harmful chemical additives [18] in today’s human society, there is a need to find a solution to reduce the adverse effects of preservatives on the human reproductive system. In spite of many reports revealing the antioxidant effect of ginger, no research has been conducted to assess the protective effects of ginger on the ovaries against SMB. This study was designed to investigate the protective effects of ginger extract on the structural changes developed in rats’ ovaries following exposure to SMB.
Materials and Methods
Animals & Experimental Design: A total of 32 virgin female Wistar rats, 12-14 weeks old weighing 220 to 250g, were obtained from the animal house of Shiraz University of Medical Sciences, Shiraz, Iran. The rats were kept under 12 hours of light-dark cycles at 21–24°C with free access to food and water. All experiments were conducted in accordance with the guiding principles for the care and use of research animals as approved by Animal Ethics Committee of the Islamic Azad University (Registration # 16030517922007).
The rats were divided into four groups of eight, consisting of, a) control group receiving distilled water, b) group 2, receiving ginger at 500 mg/kg/day, c) group 3, receiving SMB at 260 mg/kg/day, and d) combined group, receiving both SMB and ginger at the same doses used for groups 2 and 3. SMB and ginger dissolved in 1 ml of distilled water. The SMB dosage used in this study was determined based on our previous results [7]. All animals received their assigned treatment by daily gavage for 28 days. Vaginal smears were taken at regular intervals to monitor the ovarian cyclicity. When the ovaries reached their estrous stage, the animals were given their last treatment, and 24 hours later, they were euthanized with diethyl ether and their ovaries removed.
To examine the protein content and MDA in the right ovaries, the tissue samples were briefly washed and kept in liquid nitrogen. It was then weighed and stored at -70˚C until used for the experiments. The left ovaries were fixed in 4% formaldehyde buffer. Leitz Rotary Microtome was used to serially section each paraffin-embedded ovary at 26 µm in thickness. Eight to twelve sections from each ovary were selected and stained with hematoxylin and eosin [19].
Drug & Ginger Extract Preparation: The SMB stock was purchased from Sigma (EC No: 231-673-0, CAS. No: 7681-57-4; Karlsruhe, Germany) and was dissolved in distilled water at 260 mg/mL. Dried ginger rhizomes were obtained from Arsanjan grocery. The rhizomes were confirmed by Dr. Rafiee (botanist) and then powdered by grinder. After soaking the ginger powder (200 g) in one liter of 50% ethanol for 72 hours, the extraction process was initiated through percolation several times until complete exhaustion. Rotary evaporator (Genser Scientific Instruments; Bavaria, Germany) at a temperature 40-50°C was used for evaporation of the remainder solvent [20]. At the completion of this process, 20 g of the extract concentrate was obtained from a 200 g initial ginger powder.
Ovarian Volume Measurement: Ovarian volume was measured according to our previous study, using the Cavalieri principle [7]. Using a stereomicroscope, the volume of sampled sections was obtained by point counting method and the following formula 1 [21]:
1. V=Σp×(a/p)×t
where “Σp” is the total number of points superimposed on the sections; “a/p” is the area per point, and “t” is the distance between the sampled sections. Additionally, “a/p” is calculated by the following formula 2:
2. (a/p)=(Δx×Δy)/m2
where “Δx” and” Δy” are the distance between the two adjacent points on the grid in the x-axis or the y-axis, respectively. Moreover, “m” is the final linear magnification of the microscopic images.
Follicle Numbers: The stage of follicles’ maturation was determined based on the Mayer et al. method [22]. Optical disector method with Nikon E200 microscope (Nikon, Japan) with 100x magnification and the microcator (MT12, Heidenhain, Germany) was used for counting the follicle numbers. The unbiased counting frame was superimposed on the monitor images of the 26µm sections of the ovary and the oocyte nuclei were counted if they lied within the frame and did not touch the exclusion boundaries. On average, 80-100 microscopic fields were selected in each ovary via a systematic sample. The number density of different types of follicles was estimated using the following formula (3):
Nv=ΣQ/Σp.a/f.h
where “ΣQ” is the total number of the counted follicles, “h” is the tissue thickness considered for counting, “a/f” is the frame area in the true tissue scale and “Σp” is the total number of the points superimposed on the selected fields. The result of the equation was then multiplied by the total volume of the ovary to obtain the total number of follicles [23].
Lipid Peroxidation Test: The lipid peroxidation in the ovarian tissue was quantified indirectly by measuring the malondialdehyde (MDA) levels. Using thiobarbituric acid (TBA) method, the MDA concentration in homogenized tissue samples was measured at a ratio of 1/10 in 1.15% (w/v) cold KCl solution. The reaction results of MDA with TBA was the production of a pink colored complex with a maximum absorbance of 532 nm [24]. The results were presented as nmol/mg protein. The tissue protein concentration was measured by Bradford method.
Statistical Analyses: The statistical analyses were performed by one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test to determine the differences among the means, using SPSS software, v. 16. The differences in the means were considered significant at P<0.05. The data were presented as Means±SEM.
Results
Ovaries Weight & Volume: No significant changes were found in the weight of the ovaries treated with ginger or SMB compared with that of the control group. However, the ovarian tissue samples treated with SMB showed a significant decrease in the volume. Co-administration of ginger with SMB increased the ovaries’ volumes compared to the SMB treated ones, although the values did not reach that of the controls (Table 1).
The Follicle Numbers: The mean follicle numbers for the control and treatment groups are presented in Table 2.
No significant variations were observed in the ginger treated ovaries. Treatment with 260 mg/kg/day SMB caused a significant reduction in the number of primordial, primary, secondary, and graafian follicles along with a significant increase in the number of degenerate follicles (P≤0.001). Co-treatment with ginger provided protection against the adverse effects in the number of follicles exposed to SMB.
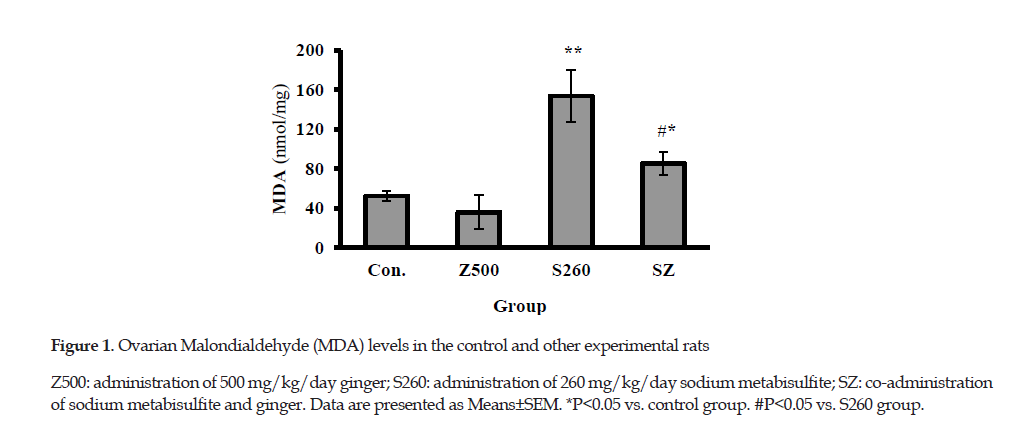
Ovarian Lipid Peroxidation: There was no significant difference in the levels of MDA for the ovaries treated with ginger compared to that of the control group. The results indicated that the ovarian MDA level significantly increased in the group treated with SMB compared to that of the control group (147.2±30.7 vs. 52.5±9.3 nmol/mg protein, P≤0.01). As shown in Figure 1, the MDA level was lower in ginger group than that in the SMB group (P≤0.05).
Discussion
In this study, rat ovarian protection against damage induced by SMB was investigated. To our knowledge, this is the first study that evaluated the protective effects of ginger against the ovarian damage induced by SMB. The structural changes in the rat’s ovary after SMB exposure were associated with reductions in the ovarian volume and follicle numbers. The co-treatment with ginger clearly restored the number of follicles close to the normal level, which is suggestive of its antioxidant property.
Over the past twenty years, the prevalence of low infertility has significantly increased [25]. It is estimated that 40%-50% of infertility cases are due to female problems [25]. There is a positive correlation between the ovarian volume and the follicles count [26]. It has been shown that reduced ovarian volume is predictive of reduced ovarian reserve in infertile women [27]. Therefore, a significant decrease in the ovarian volume and the number of mature follicles at different stages of ovulation due to SMB ingestion may reduce the chances of fertility.
Increased serum MDA, one of the end products of lipid peroxidation, is an important indicator of oxidative damage to cellular structures in humans and animals [28]. Decreases in the antioxidant production in rats treated with SMB may lead to an increase in MDA and Reactive oxygen species (ROS) and free radicals levels, which will eventually lead to a number of reproductive disorders, such as infertility [6]. It has been demonstrated that sulfite radicals are strong oxidants [5], which may interfere with the antioxidant defense system in the ovary and result in the tissue injury. The reduction in the follicular antioxidant activities could impair the scavenging efficiency against ROS, giving rise to oxidative stress [29, 30].
Increased free radicals correlate well with a low success for in vitro fertilization [31]. As reported by a previous study, the elevated intracellular ROS levels are involved in the ovarian aging [32]. Gradual decreases in the numbers of follicles and the viability of oocytes during ovarian aging lead to a gradual decline in fertility [33]. Oxidative stress is a good model for the aging process, because it has a negative impact on the oocytes’ development in young mice [34]. In fact, oxidative stress will finally leads to pathological ovarian aging through a decline in the antioxidant defense and a rise in the ROS production [33]. In this study, the MDA level increased in the SMB treated rats as compared to that in the control group. The oxidative stress induced by the overproduction of free radicals apparently played a major role in the development of the ovarian damages subsequent to sulfite treatment. Further, decreased MDA level was observed in the rats treated with combined SMB and ginger, which is suggestive of the inhibition of the oxidative activities in the presence of ginger. This finding is in agreement with a previous study suggesting that ginger significantly lowered the lipid peroxidation [35]. This effect of ginger is believed to occur by the induction or enhancement of antioxidant enzymes [14].
Recommendation for Future Studies: Stereological examination of ovary showed an increase in the degenerate follicles. The measurement of serum estrogen and antioxidant enzymes levels helped confirm the results. Therefore, we recommend that researchers consider these variables in the future studies.
Conclusions
This study demonstrated that SMB at 260 mg/kg stimulated lipid peroxidation and reduced the number of ovarian follicles at various stages of ovulation in rats. With structural changes in the rat ovary, SMB appears to be effective on the female rat fertility. On the other hand, ginger restored the number of follicles towards normalcy and reduced the ovarian tissue MDA levels. Therefore, the protective effect of ginger against the ovarian damages subsequent to sulfite exposure might be mediated through the inhibition of the scavenging activity of free radicals. Therefore, women are likely to be at increased risk of ovarian damages and fertility disorders as a result of dietary exposure to sulfites from processed foods and beverages. We recommend that women who consume processed foods and beverages add ample ginger to their daily diets.
Ethical Considerations
Compliance with ethical guidelines
All experiments were conducted in accordance with the “Guiding Principles for the Care and Use of Research Animals” as approved by the Animal Ethics Committee of Islamic Azad University, Arsanjan, Iran.
Funding
The present paper was extracted from the M.Sc. thesis project conducted by Mrs. Nahid Rezaee.
Author's contributions
All authors were equally contributed in preparing this article.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
We gratefully thank Mr. Kuhpeyma for his valuable technical assistance provided to this project.
References
1.Ozturk N, Yargicoglu P, Derin N, Akpinar D, Agar A, Aslan M. Dose-dependent effect of nutritional sulfite intake on visual evoked potentials and lipid peroxidation. Neurotoxicol Teratol. 2011; 33(2):244-54. [DOI:10.1016/j.ntt.2010.09.002] [PMID]
2.Vally H, Misso NL. Adverse reactions to the sulphite additives. Gastroenterol Hepatol Bed Bench. 2012; 5(1):16-23. [PMID] [PMCID]
3.Elmas O, Aslan M, Cağlar S, Derin N, Ager A, Alicigüzel Y, et al. The prooxidant effect of sodium metabisulfite in rat liver and kidney. Regul Toxicol Pharmacol. 2005; 42(1):77-82. [DOI:10.1016/j.yrtph.2005.01.010] [PMID]
4.Vally H, Misso NLA, Madan V. Clinical effects of sulphite additives. Clin Exp Allergy. 2009; 39(11):1643-51. [DOI:10.1111/j.1365-2222.2009.03362.x] [PMID]
5.Ranguelova K, Rice AB, Lardinois OM, Triquigneaux M, Steinckwich N, Deterding LJ, et al. Sulfite-mediated oxidation of myeloperoxidase to a free radical: Immuno-spin trapping detection in human neutrophils. Free Radic Biol Med. 2013; 60:98-106. [DOI:10.1016/j.freeradbiomed.2013.01.022] [PMID] [PMCID]
6.Agarwal A, Aponte-Mellado A, Premkumar BJ, Shaman A, Gupta S. The effects of oxidative stress on female reproduction: a review. Reprod Biol Endocrinol. 2012; 10:49. [DOI:10.1186/1477-7827-10-49] [PMID] [PMCID]
7.Rezaee N, Nematollahi Z, Shekarfroush S, Hoseini E. Effect of sodium metabisulfite on rat ovary and lipid peroxidation. Iran J Toxicol. 2016; 10(2):23-8. http://ijt.arakmu.ac.ir/article-1-431-en.html
8.Bolon B, Bucci TJ, Warbritton AR, Chen JJ, Mattison DR, Heindel JJ. Differential follicle counts as a screen for chemically induced ovarian toxicity in mice: Results from continuous breeding bioassays. Fundam Appl Toxicol. 1997; 39(1):1-10. [DOI:10.1006/faat.1997.2338] [PMID]
9.Hoyer PB, Keating AF. Xenobiotic effects in the ovary: Temporary versus permanent infertility. Expert Opin Drug Metab Toxicol. 2014; 10(4):511-23. [DOI:10.1517/17425255.2014.880690] [PMID]
10.Varalakshmi B, Anand V, Kumar S. In vitro biomolecular protective effect of Cinnamomum zeylanicum bark extracts against oxidative damage. Int J Pharm Pharm Sci. 2016; 8(7):369-72. https://innovareacademics.in/journals/index.php/ijpps/article/view/12205
11.Lü JM, Lin PH, Yao Q, Chen C. Chemical and molecular mechanisms of antioxidants: Experimental approaches and model systems. J Cell Mol Med. 2010; 14(4):840-60. [DOI:10.1111/j.1582-4934.2009.00897.x] [PMID] [PMCID]
12.Chrubasik S, Pittler MH, Roufogalis BD. Zingiberis rhizoma: A comprehensive review on the ginger effect and efficacy profiles. Phytomedicine. 2005; 12(9):684-701. [DOI:10.1016/j.phymed.2004.07.009] [PMID]
13.Akbari A, Nasiri K, Heydari M, et al. The protective effect of hydroalcoholic extract of zingiber officinale roscoe (ginger) on ethanol-induced reproductive toxicity in male rats. J Evid Based Complementary Altern Med. 2017; 22(4):609-17. [DOI:10.1177/2156587216687696] [PMID] [PMCID]
14.Ahmed RS, Seth V, Banerjee BD. Influence of dietary ginger (Zingiber officinales Rosc) on antioxidant defense system in rat: Comparison with ascorbic acid. Indian J Exp Biol. 2000; 38(6):604-6. [DOI:10.1016/S0278-6915(00)00019-3] [PMID]
15.Yilmaz N, Seven B, Timur H, Yorgancı A, Inal HA, Kalem MN, et al. Ginger (zingiber officinale) might improve female fertility: A rat model. J Chin Med Assoc. 2018; 81(10):905-11. [DOI:10.1016/j.jcma.2017.12.009] [PMID]
16.Rhode J, Fogoros S, Zick S, Wahl H, Griffith KA, Huang J, et al. Ginger inhibits cell growth and modulates angiogenic factors in ovarian cancer cells. BMC Complement Altern Med. 2007; 7:44. [DOI:10.1186/1472-6882-7-44] [PMID] [PMCID]
17.Amin A, Hamza AA. Effects of Roselle and Ginger on cisplatin-induced reproductive toxicity in rats. Asian J Androl. 2006; 8(5):607-12. [DOI:10.1111/j.1745-7262.2006.00179.x] [PMID]
18.Ginode AG, Pimparkar KM, Nawkar MS, Deshpande YN. A Survey study on Viruddha Aahar (Fast food) Consumption Amongst College Students. Int J Ayurvedic Med. 2018; 9(3):157-66. https://ijam.co.in/index.php/ijam/article/view/1137#
19.Karbalay-Doust S, Noorafshan A. Stereological estimation of ovarian oocyte volume, surface area and number: Application on mice treated with nandrolone decanoate. Folia Histochem Cytobiol. 2012; 50(2):275-9. [DOI:10.5603/FHC.2012.0037] [PMID]
20.Shalaby M, Mouneir SM. Effect of zingiber officinale roots and cinnamon zeylanicum bark on fertility of male diabetic rats. Global Veterinaria. 2010; 5(6):341-7. https://www.idosi.org/gv/gv5(6)10/8.pdf
21.Charleston JS, Hansen KR, Thyer AC, Charleston LB, Gougeon A, Siebert JR, et al. Estimating human ovarian non-growing follicle number: The application of modern stereology techniques to an old problem. Hum Reprod. 2007; 22(8):2103-10. [DOI:10.1093/humrep/dem137] [PMID]
22.Myers M, Britt KL, Wreford NGM, Ebling FJP, Kerr JB. Methods for quantifying follicular numbers within the mouse ovary. Reproduction. 2004; 127(5):569-80. [DOI:10.1530/rep.1.00095] [PMID]
23.Mehranjani MS, Noorafshan A, Hamta A, et al. Effects of vitamin E on ovarian tissue of rats following treatment with p-nonylphenol: A stereological study. Iran J Reprod Med. 2010; 8(1):1-9. http://journals.ssu.ac.ir/ijrmnew/article-1-172-en.html&sw=Vitamin+E.
24.Botsoglou NA, Fletouris DJ, Papageorgiou GE, Vassilopoulos VN, Mantis AJ, Trakatellis AG. Rapid, sensitive, and specific thiobarbituric acid method for measuring lipid peroxidation in animal tissue, food, and feedstuff samples. J Agric Food Chem. 1994; 42(9):1931-7. [DOI:10.1021/jf00045a019]
25.Showell MG, Brown J, Clarke J, et al. Antioxidants for female subfertility. Cochrane Database Syst Rev. 2013; CD007807. [DOI:10.1002/14651858.CD007807.pub2]
26.Kelsey TW, Wallace WHB. Ovarian volume correlates strongly with the number of nongrowing follicles in the human ovary. Obstet Gynecol Int. 2012; 2012:305025. [DOI:10.1155/2012/305025] [PMID] [PMCID]
27.Bowen S, Norian J, Santoro N, Pal L. Simple tools for assessment of ovarian reserve (OR): Individual ovarian dimensions are reliable predictors of OR. Fertil Steril. 2007; 88(2):390-5. [DOI:10.1016/j.fertnstert.2006.11.175] [PMID] [PMCID]
28.Negre-Salvayre A, Coatrieux C, Ingueneau C, Salvayre R. Advanced lipid peroxidation end products in oxidative damage to proteins. Potential role in diseases and therapeutic prospects for the inhibitors. Br J Pharmacol. 2008; 153(1):6-20. [DOI:10.1038/sj.bjp.0707395] [PMID] [PMCID]
29.Tatone C, Amicarelli F, Carbone MC, Monteleone P, Caserta D, Marci R, et al. Cellular and molecular aspects of ovarian follicle ageing. Hum Reprod Update. 2008; 14(2):131-42. [DOI:10.1093/humupd/dmm048] [PMID]
30.Carbone MC, Tatone C, Delle Monache S, Merci R, Caserta D, Colonna R, et al. Antioxidant enzymatic defences in human follicular fluid: Characterization and age-dependent changes. Mol Hum Reprod. 2003; 9(11):639-43. [DOI:10.1093/molehr/gag090] [PMID]
31.Wiener-Megnazi Z, Vardi L, Lissak A, Shnizer S, Reznick Z, Ishai D, et al. Oxidative stress indices in follicular fluid as measured by the thermochemiluminescence assay correlate with outcome parameters in in vitro fertilization. Fertil Steril. 2004; 82 Suppl 3:1171-6. [DOI:10.1016/j.fertnstert.2004.06.013] [PMID]
32.Tarin JJ. Potential effects of age-associated oxidative stress on mammalian oocytes/embryos. Mol Hum Reprod. 1996; 2(10):717-24. [DOI:10.1093/molehr/2.10.717] [PMID]
33.Li Q, Geng X, Zheng W, Tang J, Xu B, Shi Q. Current understanding of ovarian aging. Sci China Life Sci. 2012; 55(8):659-69. [DOI:10.1007/s11427-012-4352-5] [PMID]
34.Zhang X, Wu XQ, Lu S, Guo YL, Ma X. Deficit of mitochondria-derived ATP during oxidative stress impairs mouse MII oocyte spindles. Cell Res. 2006; 16(10):841-50. [DOI:10.1038/sj.cr.7310095] [PMID]
35.Morakinyo AO, Adeniyi OS, Arikawe AP. Effects of Zingiber officinale on reproductive functions in the male rat. Afr J Biomed Res. 2008; 11(3):329-34. [DOI:10.4314/ajbr.v11i3.50750]
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |