BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
URL: http://ijt.arakmu.ac.ir/article-1-821-en.html


 1, Benjamin O. Emikpe2
1, Benjamin O. Emikpe2 

 , Ademola A. Oyagbemi3
, Ademola A. Oyagbemi3 

 , Theophilus A. Jarikre4
, Theophilus A. Jarikre4 

 , Clement O. Adjei1
, Clement O. Adjei1 

2- Department of Pathobiology, School of Veterinary Medicine, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana.
3- Department of Veterinary Physiology and Biochemistry, Faculty of Veterinary Medicine, University of Ibadan, Nigeria.
4- Department of Veterinary Pathology, Faculty of Veterinary Medicine, University of Ibadan, Nigeria.
Introduction
The kidneys are involved in various important functions, such as maintenance of proper ion levels in serum, regulation of acid-base homeostasis, and the removal of nitrogenous wastes. Through renal impairments, these functions are disrupted and there occurs accumulation of waste products, including urea, creatinine and other ions in the blood [1]. It is estimated that the incidence of chronic kidney diseases is 10%-13% globally [2]. A major cause of renal dysfunction and damage is the effect of nephrotoxins. These include therapeutic drugs, such as antibiotics, chemotherapeutic drugs, and various industrial chemicals and heavy metals [3]. Two of the most highly documented nephrotoxic drugs are gentamicin and cisplatin, which primarily induce injury to the proximal convoluted tubules of the kidneys [1, 4].
Aminoglycosides are either naturally occurring or semi-synthetic agents. Their basic structure consists of two or more aminosugars joined by a glycosidic linkage to an aminocyclitol ring [5]. Gentamicin and kanamycin are typical examples of aminoglycosides. The major problem associated with aminoglycosides is their toxicity to the kidneys, causing renal dysfunction [6]. About 10%-20% of people who take gentamicin experience renal disorders [1]. Gentamicin increases the Reactive Oxygen Species (ROS) levels while reducing that of antioxidants and inhibitory effects, including glutathione peroxidase. Nephrotoxicity associated with gentamicin is characterized by increased serum creatinine and urea nitrogen, necrosis of the proximal tubules, and tubular epithelial edema [6].
Cisplatin (Cisp) is another damaging drug to the kidneys. It is widely applied to the treatment of various cancer tumors and is one of the most effective drugs used in chemotherapy. Unlike most other cancer drugs, Cisp is a simple inorganic platinum-based compound [7]. It is known for its ability to crosslink DNA, inhibiting DNA replication and synthesis, and kills the rapidly dividing cancer cells [8]. Two major limitations of Cisp are its drug resistance and the lethal effects on normal body tissues, such as toxicity to nerves, kidneys and ears. Cisplatin’s toxicity on the kidneys is usually observed after ten days of administration, manifested with a decline in the glomerular filtration rate (GFR), increased serum creatinine, hypomagnesemia and hypokalaemia [8]. This appears to be due to the increased concentration of free radicals in the tissues exposed to this drug [4].
Interest in medicinal plant research and application has risen due to the many reported side effects of synthetic drugs and their relatively high cost [9]. About 80% of natives in developing countries rely on traditional medicine, especially plant-based drugs, for their primary health care [10]. Traditionally, herbs have been used for the treatment of renal disorders induced by drugs and numerious toxins. In this context, many medicinal plants have been tested for their nephroprotective effects in nephrotoxicities induced by GM and Cisp [11-14]. Medicinal plants are used as curatives due to their active components with medicinal value. These components, i.e., phytochemicals, act through mechanisms similar to those well studied for synthetic drugs [15].
Acalypha wilkesiana Müll. Arg., ‘inferno’ (Euphorbiaceae family) is a commonly cultivated ornamental plant marketed as a hedge. It is called the copperleaf and widely distributed in the tropics. Few pharmacological and phytochemical assessments have been done on this plant. According to Larbie et al. [16], the hydroethanolic extract of this plant has been shown to contain triterpenoids, alkaloids, flavonoids, glycosides, coumarins, sterols, and hydrolyzable tannins. That study also revealed high antioxidant and significant bactericidal activities in A. wilkesiana. This plant has also been demonstrated to improve the clearance of blood glucose and is toxicologically safe in animal models [17, 18]. Currently, the acute and subacute hepatoprotective activity of this plant is under evaluation [19]. However, the nephroprotective effect has not been reported. These studies have referred to this plant as Acalypha inferno. Previously, A. indica root extract was evaluated for its protective effect in high-fructose and high-cholesterol diet-fed rats [20], which was found to be effective. Similarly, A. wilkesiana has been evaluated in cyclophosphamide-induced toxicity [21]. These are related to the extract being evaluated by us. The present study is an extension to the aforementioned research, aimed at investigating the nephroprotective effect of A. wilkesiana against renal damage caused by GM and Cisp in rats, based on the biochemical, histological and immunohistochemical assessments.
Materials and Methods
Plant Preparation and Extraction: The A. wilkesiana leaves were collected and prepared as previously described [16-19]. Briefly, the leaves were identified as that of A. wilkesiana ‘inferno’ (voucher number KNUST/HM/2017/L018), air-dried under shade and powdered. To prepare the 50% hydroethanolic extract, 100g of the powder was suspended in 1L of 50% ethanol (50:50 v/v ethanol: water). The mixture was agitated on a mechanical shaker for 24 hours at room temperature (22-25°C). The solution was then filtered and concentrated in a Heidolph Rotary Evaporator (Germany) at 60℃ under vacuum. The extract was freeze-dried (Labconco, England) to obtain the A. wilkesiana hydroethanolic leaf extract for use in this study. The lyophilized material was reconstituted in normal saline to prepare the respective doses of the extract.
Animals: Thirty six male Wistar albino rats, purchased from the animal facility of the University of Ghana School of Medical Sciences, Accra, Ghana, were used for the experiments. The animals were transferred to the holding facility of the Department of Biochemistry and Biotechnology, KNUST and kept in aluminium cages bedded with wood shavings. The animals were allowed to acclimatize to laboratory conditions for 2 weeks. Throughout the study, optimum laboratory conditions were maintained at 24-26℃ and 40%-70% relative humidity. The animals had free access to standard rat feed (Mash, AGRICARE, Kumasi, Ghana) and tap water ad libitum except in an overnight fast before the first treatment and before sacrifice. The animals were identified by marking them on the tails with permanent markers. This study was conducted based on the guidelines set by the Committee for the Purpose of Control and Supervision of Experiment in Animals (CPCSEA, New Delhi, India) and the Guide for the Care and Use of Laboratory Animals [22]. All animals were humanely handled during the experiments. In addition, the study protocol was reviewed and approved by a veterinarian on the research team. For both experimental model (GM or Cisp), the rats were divided into nine groups of four per group.
Drug-induced Nephrotoxicity: Gentamicin (GM, Letpa Pharmaceuticals Limited, Accra, Ghana; 120 mg/kg body weight, administered intramuscularly) and cisplatin (Cisp, Celon Labs, India; 7 mg/kg administered intraperitoneally) were used to induce renal injury in the experimental animals. GM was administered from the 2nd to 7th days of the experiment while Cisp was administered on the 3rd day according to the procedure described by Chatterjee et al. [23] with the previously used modifications [14].
Experimental Design: In both GM and Cisp experimental models (N=36), animals in Group I served as normal controls and were orally administered with 1.0 mL/kg normal saline each day. Animals in Group II were treated with nephrotoxins only, group III animals received the standard drug (silymarin, 120 mg/kg) alongside the nephrotoxins while animals in groups IV and V were treated with 100 mg/kg or 250 mg/kg of the extract, respectively, plus either nephrotoxin.
For the GM experiment (N=16), Group II animals were treated with GM from days 2-7 and maintained as GM control. Animals in Group III were pre-treated with 120 mg/kg silymarin from days 1-10 concomitantly treated with GM from days 2 to 7. Groups IV and V were treated daily with 100 mg/kg or 250 mg/kg of the extract, respectively, from days 1-10 by oral route. Additionally, they were treated with GM on days 2-7.
In the Cisp experiment (N=16), animals in groups II to V were treated with an intraperitoneal injection of 7.0 mg/kg of Cisp on day 3. Rats in Group II were maintained as Cisp controls while those in group III were treated with 120 mg/kg of silymarin on days 1-10. Groups IV and V were given daily oral doses of 100 mg/kg or 250 mg/kg of the extract, respectively, for 10 days plus a dose of Cisp given on day 3. The animals were weighed prior to and at the end of the experiment for both experimental models. All animal treatments lasted 10 days regardless of the groups.
Effect of Treatment on Hematological and Biochemical Parameters: Animals were fasted overnight after the experiment on day 10 and sacrificed on day 11 by light ether anesthesia. The fur from the neck region was removed, the jugular vein exposed, and cut with a sterile surgical blade. An aliquot of venous blood was collected into EDTA tubes for hematological analysis, using the Sysmex Hematology System (USA). The standard parameters measured included white blood cells, red blood cells, hemoglobin, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, platelet, lymphocytes, neutrophils, red blood cell distribution width, red blood cell distribution width – corpuscular volume, platelet distribution width, mean platelet volume, platelet large cell ratio, and plateletcrit.
Three mL of blood samples were dispensed into gel-activated tubes for biochemical parameters. The blood was allowed to clot then centrifuged at 3000 rpm for 5 minutes (Eppendorf Centrifuge 5804). The serum was obtained in appropriate tubes for biochemical analyses, using the Selectra E analyser from Vital Scientific (Tokyo, Japan) with the reagents purchased from ELITECH (Puteaux, France). The following serum parameters were analyzed: ALT (alanine aminotransferase), Urea, Creatinine, sodium, potassium, and chloride.
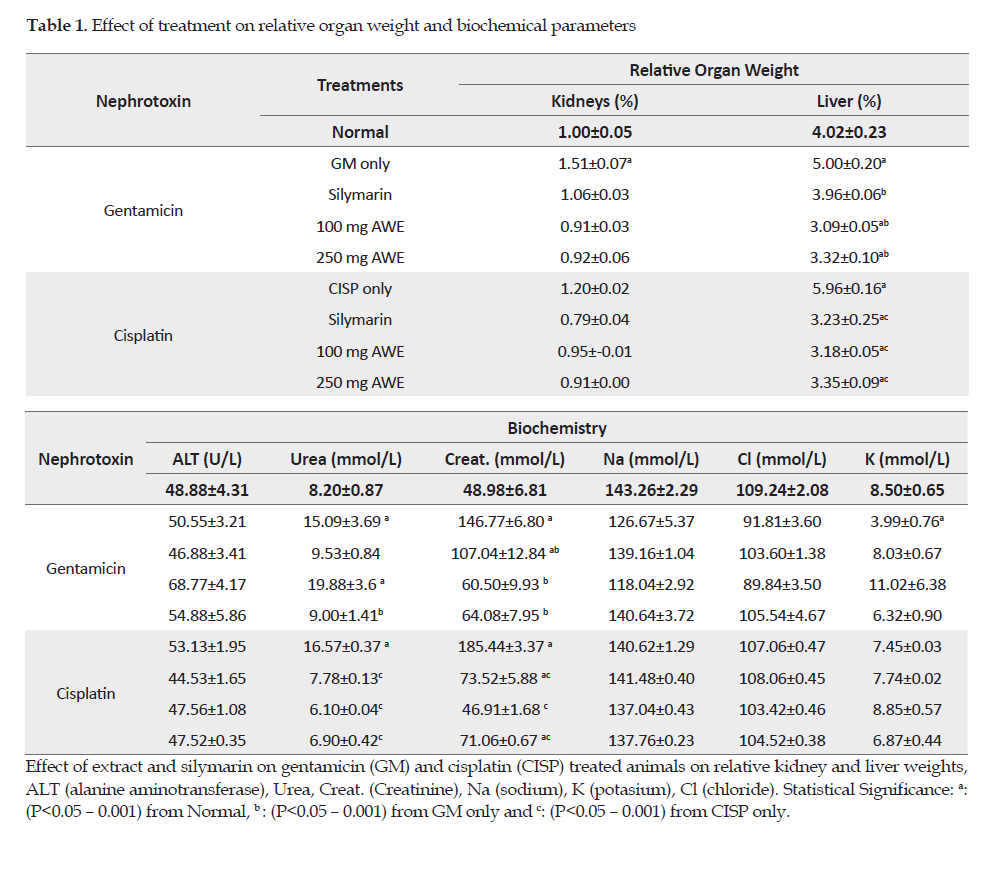
Effect of Treatments on Organ Weight: The kidneys and livers from the sacrificed rats were excised, cleared of fat, and washed with normal buffered saline. The kidneys were blotted to dryness with a clean tissue, weighed and the absolute organ weight was recorded for each tissue sample. The relative organ weight for each rat was calculated using the following formula:
Histology & Kidney Antioxidant Assays: The left kidney from each rat was preserved in 10% formalin and processed for histological assessments. The kidney sections were then stained using haematoxylin and eosin (H&E). Photomicrographs were obtained and analysed by two independent pathologists. The right kidney from each rat was also kept in phosphate-buffered saline. Kidney tissues were separately homogenized in 10 mL of 100 mM KH2PO4 buffer which contained 1 mM EDTA at pH 7.4. This was centrifuged at 12,000 rpm for 30 minutes at 4°C. The supernatants were collected and assayed for the kidney pro- and anti-oxidants parameters, including total protein [24], superoxide dismutase (SOD) [25], malondialdehyde (MDA) [26], reduced glutathione (GSH) [27], glutathione peroxidase (GPx) [28], glutathione transferase (GST) [29], nitric oxide [25] and hydrogen peroxide (H2O2) [30].
Immunohistochemistry for expression of pro-inflammatory markers: Streptavidin-biotin peroxidase complex kit (M IHC Select Detection System, HRP/DAB, Merck, Germany, Lot: 2775482) was used for the immunohistochemical staining, the procedure of which described by Jarikre and Emikpe [31] and also used previously [14]. The primary antibodies were interleukin (IL) 17 (SAB3701439, Lot: R127357), monoclonal mouse cyclooxygenase (COX) antibody (sc-19999, Lot # L1113), and interleukin IL 23 (Cat. #06-1079, Lot # 2914943). An Amscope MU900 digital camera (California, USA) attached to a microscope was used to take the photomicrographs. Images were then quantified for the intensity of staining, using the reciprocal intensity of the stained markers (COX-2, IL17 and IL23) on open source Fiji software (ImageJ). The optic density (OD) was calculated using the following formula:
A value of “50” was used as the cut-off point for the OD. The grading for immune-positive staining was as follows: weak (50-100), moderate (101-200), and strong (>200).
Data analyses: The results were expressed as Means± Standard Error of the Mean±SEM and the differences among the means were calculated, using 1-way analyses of variance (AVOVA), followed by Tukey’s post hoc test. The data were also analyzed, using GraphPad Prism 7 (GraphPad Software Inc., CA, USA). All analyses were conducted assuming a significance value of P≤0.05.
Results
Effect of Treatment on Organ Weights: Table 1 shows the effect of treatment on kidneys and liver weights. The administration of GM and Cisp resulted in significant increases in the relative weights of the kidneys and liver. Co-administration with silymarin and the extract at 100 mg or 250 mg/kg prevented significant variations in the organ weights.
Effect of Treatment on Serum Biochemical Parameters: GM and Cisp treatments resulted in significant increases in the serum levels of urea and creatinine. The electrolytes were, however, not affected. Extract treatment restored such increases to near-normal levels (Table 1).
Effect of Treatment on Haematological Parameters: Table 2 presents the effect of treatment on the hematological parameters for the normal controls and the treated animals. The acute administration of GM and Cisp did not have any effect.
All animals showed normal levels of the respective parameters compared to those of the normal controls except for significant increases (P<0.05-0.01) in the platelet counts with GM and Cisp treatments. Silymarin and the extract co-treatment caused significant decreases in the platelets counts (P<0.05-0.01) particularly in the Cisp treated group.
Effect of Treatment on the Kidneys’ Pro- and Antioxidant Parameters: The GM administration resulted in significant decreases in the levels of glutathione transferase (GST) and peroxidase (GPx). The Cisp treatment resulted in significant decreases in GST and GPx levels and an increase in H2O2 levels (Table 3).
Silymarin and the extract treatment restored these parameters to near normal. Significantly, the levels of GPx, SOD and GST were increased while the MDA and NO levels were reduced in rats co-treated with GM, silymarin and the extract. In Cisp treatment group, only the GST levels were significantly increased.
Effect of Treatment on Histopathology of Rats’ Kidneys: Group I rats (normal controls) did not show any damage in the tubular epithelial tissue, i.e., the parenchyma cells had normal features (Plate 1A).
In the GM-treated kidneys, there were glomerular tuft atrophy, tubular epithelial degeneration, coagulation, necrosis, interstitial edema and lymphocyte infiltration (Plate 1B). The co-treatment with silymarin and the extract demonstrated a reversal of these lesions in the kidney tissues except for the presence of a few patchy tubular coagulative necrosis and renal tubular ectasia (Plates 1C to 1E). Also, the Cisp treatment induced tubular epithelial degeneration and necrosis with casts detected in the tubular lumen, disruption of the basement membrane (tubulorhexis) and extensive inflammation (Plate 1F). Treatment with silymarin and the extract showed a reversal of the lesions except for a few areas of tubular epithelial necrosis, tubular ectasia of the lumen with casts and inflammation at 100 mg/kg of the extract (Plates 1G to 1H) but no observable lesion at 250 mg/kg of the extract (Plate 1I).
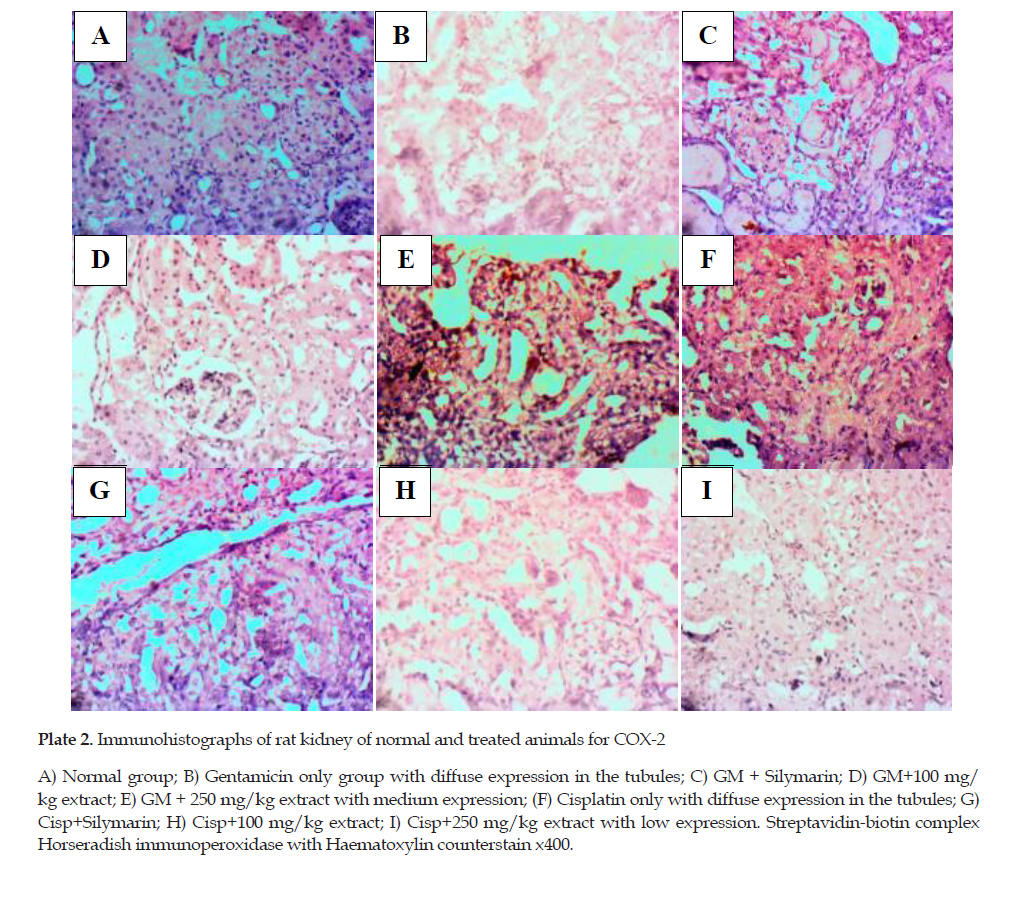
Effect of Treatments on Liver Pro-inflammatory Cytokines in the Rat Kidneys: There was minimal expression of COX-2 in group I rats (Plate 2A) while a strong expression of COX-2 was observed in the glomerular and proximal convoluted tubular cells in response to the GM treatment (Plate 2B) and the Cisp treatment (Plate 2F) only.
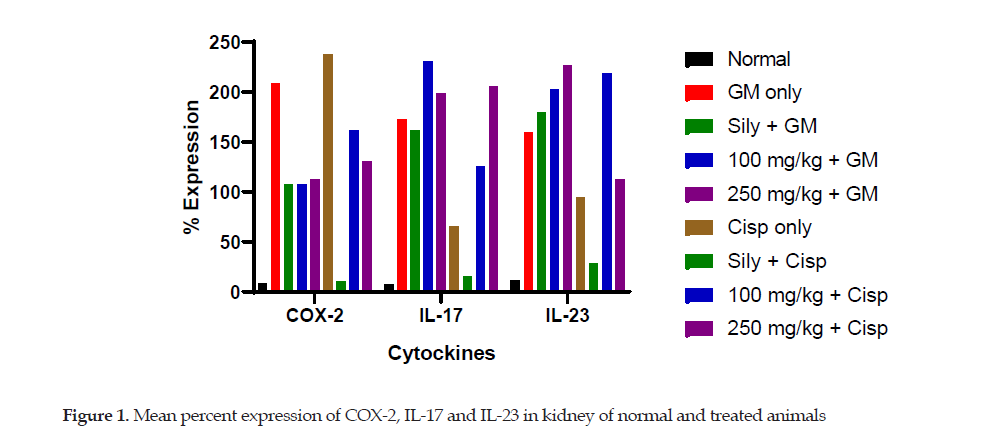
However, the COX-2 expression was reduced remarkably after treatment with silymarin and moderately with the extract at varying doses against the toxicity of both GM (Plates 2C to 2E) and Cisp (Plates 2G to 2I). The expressions of IL-17 (Plates 3A to 3I) and IL-23 (Plates 2I to 4A) were not remarkably modulated by the treatment with the extract (Figure 1).
Discussion
Exposure of the kidneys to nephrotoxins may result in structural and functional injuries in them [1]. Morphological, biochemical, and hematological aberrations are observed during kidney injuries, resulting in increased levels of electrolytes, serum creatinine, urea, and total protein [6]. Red blood cell production is reduced in renal damage due to the decreased erythropoietin synthesized by the kidneys [32]. Red cell indices (HCT, MCH, MCHC, HGB, etc.) are also reduced in renal damages [33]. Morphological changes associated with kidney injuries include severe proximal tubular necrosis, glomerular atrophy and extensive tubular epithelial edema [6].
Gentamicin is one of the most used aminoglycoside antibiotics in the treatment of Gram-negative bacterial infections. Manshare et al. [1] report that between 10%-20% of individuals treated with GM suffer renal injuries, which are the major complications of this drug. Oxidative stress, as evident by the elevated levels of ROS, increased lipid peroxidation and NO synthesis, decreased GST, and reduced activity of catalase, superoxide dismutase and glutathione peroxidise, is believed to underlie the GM nephrotoxicity [6]. Cisplatin is a platinum-based drug used for its anticancer properties. The increased resistance to Cisp and the lethal effects on bodily tissues are the two major limitations of Cisp. The nephrotoxicity of Cisp is associated with reduced GFR, increased serum creatinine, hypomagnesemia and hypokalemia [8]. Cisp-induced renal damage is also attributed to its ability to induce oxidative stress [4].
Consistent with the results reported by Ghaznavi et al. [6] and Shelkea et al. [34], GM and Cisp administration at 120 mg/kg and 7 mg/kg, respectively, resulted in a significant increase in the serum creatinine and urea levels compared those seen in the normal controls. Since a major kidneys’ function is the excretion of nitrogenous waste, such as creatinine and urea, structural injuries to the kidneys result in the accumulation of these metabolites in the blood [1]. The co-administration of the extract at 100mg or 250 mg/kg or silymarin significantly reduced the serum creatinine levels as compared to the results for treatment with either Cisp or GM only. The urea levels were considerably reduced by the extract at 250mg in comparison with that for GM alone.
However, the reduction in the serum urea was not significant in the Cisp-induced nephrotoxicity. Administration of the extract at 100 mg did not alter the urea levels in response to the GM-induced nephrotoxicity. All other biochemical parameters measured were not affected by the treatments. Larbie et al. [16] demonstrated that the hydroethanolic extract of A. wilkesiana had high levels of antioxidants, flavonoids and hydrolyzable tannin activities. The oxidative stress induced by Cisp and GM normally results in kidney damage as evident by increases in serum creatinine and urea. The extract’s antioxidant properties may have reversed or prevented the increases in these parameters in our study. Again, our results revealed that the extract at 250mg/kg was better at reversing this increase compared to that noted for the100 mg/kg. The observed increases in the levels of GST and GPx, and decreases in MDA and NO confirm the antioxidant effects of the extracts. The protective effect of A. indica [20] and the related species, A. wilkesiana [21] appears to occur via such mechanisms as antioxidant and improved mitochondrial permeability.
The biochemical changes in both the serum and kidney homogenates following the administration of GM or Cisp was corroborated by the abnormal architectural changes found upon the histologic examinations of the kidneys. These included glomerular atrophy, tubular epithelial coagulative necrosis and cellular infiltrates (inflammation), tubular basement disruption (tubulorrhexis), and multiple foci of tubular casts. The co-administration of the extract at 100 or 250mg/kg reversed these abnormal alterations to near normal with less severity especially at 250mg/kg of the extract. These changes were also underscored by the expression of COX-2 in the renal tubular epithelium, especially as its expression was ameliorated with the increased concentration of the extract.
Inflammation has been demonstrated to be a predominant cause of parenchymal cell death in the kidney and severe cases of acute kidney injuries [35], including acute injuries following nephrotoxins administration. This inflammatory response may be mediated by two different but related, arms of the immune system: both innate and adaptive immunity. The current study demonstrated excessive expression of pro-inflammatory cytokine (COX-2), IL-17 and IL-23) in the renal parenchyma following GM or Cisp treatment. The co-administration with the extract moderately but significantly down-regulated the COX-2 expression. Cyclooxygenase, also known as prostaglandin endoperoxidase synthase, is a key enzyme in the conversion of arachidonic acid to prostaglandins, with two known isoforms of COX-1 and COX-2 [36]. The over-expression of COX-2 has been reported in inflammation and renal dysfunction.
The role of IL-17A in the pathogenesis of acute and chronic kidney disease has been demonstrated [37] and that its blockade reduces albuminuria and kidney injury in diabetic nephropathy [38]. Interleukin-23 is a pro-inflammatory cytokine, playing an important bridging role between the innate and adaptive immune responses [39]. The supportive data coming from murine models, suggesting a positive effect where blocking IL-23 ameliorates kidney and skin diseases [40]. In the current study, the treatment with the extract did not lead to significant down-regulation in the expression of IL-17 and IL-23, ruling-out any possible role of the extract’s protective effect through this mechanism. The current study was limited to hematological, biochemical and some proinflammatory cytokines. Down regulation with respect to IL-17 and IL-23 did not seem to be related to the protective effect of the extract. Thus other mechanisms could be explored to confirm the protective effect and support a possible translation of the outcomes to human subjects.
Conclusion
The current study demonstrated that the cultivated ornamental hedge, A. wilkesiana ‘inferno’ has protective effects against kidney damages induced by gentamicin and cisplatin by modulating the biochemical parameters, improving kidney micro-structures, modulating pro-oxidant and antioxidant properties (NO versus GST & GPx), and down-regulating the expression of COX-2. The extract can, therefore, be considered as an alternative and effective agent used in the management of kidney diseases, which is likely to be developed into a potent drug for clinical applications.
Ethical Considerations
Compliance with ethical guidelines
All animals were humanely treated. The study protocol was reviewed and approved by a veterinarian on the research team.
Funding
The current study did not receive any funding from local, national or international agency be it for proft or non-proft.
Author's contributions
Conceptualization and Methodology: Christopher Larbie, Benjamin O. Emikpe, Ademola A. Oyagbemi, Theophilus A. Jarikre; Formal analysis & Investigations: Christopher Larbie, Benjamin O. Emikpe, Ademola A. Oyagbemi, Clement O. Adjei; Writing of the original draft preparation, review & editing: Christopher Larbie, Clement O. Adjei. All authors approved the final draft of the manuscript prior to submission.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgements
The technical assistance of Raymond Adocta Bukari, and Mr. I.C. Mbapan are highly appreciated.
References
1.Manshare K, Anand A, Mahajan S, Satija S, Sharma N, Khurana N, et al. Evaluation of nephroprotective activity of gallic acid in gentamicin-induced rat model of nephrotoxicity. Int J Green Pharm. 2018; 2018:48-52. [DOI:10.22377/ijgp.v12i02.1829]
2.Hill NR, Fatoba ST, Oke JL, Hirst JA, O'Callaghan CA, Lasserson DS, et al. Global prevalence of chronic kidney disease - A systematic review andmeta-Analysis. PLoS One. 2016; 11(7):e0158765. [DOI:10.1371/journal.pone.0158765] [PMID] [PMCID]
3.Kovacic P, Sacman A, Wu-Weis M. Nephrotoxins: Widespread role of oxidative stress and electron transfer. Curr Med Chem. 2002; 9(8):823-47. [DOI:10.2174/0929867024606803] [PMID]
4.Karasawa T, Steyger PS. An integrated view of cisplatin-induced nephrotoxicityand ototoxicity. Toxicol Lett. 2015; 237(3):219-27. [DOI:10.1016/j.toxlet.2015.06.012] [PMID] [PMCID]
5.Martínez-Salgado C, López-Hernández FJ, López-Novoa JM. Glomerularnephrotoxicity of aminoglycosides. Toxicol Appl Pharmacol. 2007; 223(1):86-98. [DOI:10.1016/j.taap.2007.05.004] [PMID]
6.Ghaznavi H, Fatemi I, Kalantani H, Taghi Hosseini Tabatabaei SM, Mehrabani M, Kalantar M, et al. Ameliorative effects of gallicacid on gentamicin-induced nephrotoxicity in rats. J Asian Nat Prod Res. 2018; 20(12):1182-93 [DOI:10.1080/10286020.2017.1384819] [PMID]
7.Gelasco A, Lippard SJ. Anticancer activity of cisplatin and related complexes. In: Clarke MJ, Sadler PJ, editors. Metallopharmaceuticals I. Topics in Biological Inorganic Chemistry. Berlin: Springer; 1999. [DOI:10.1007/978-3-662-03815-4_1]
8.Pabla N, Dong Z. Cisplatin nephrotoxicity: Mechanisms and renoprotective strategies. Kidney Int. 2008; 73(9):994-1007. [DOI:10.1038/sj.ki.5002786] [PMID]
9.Karagoz ID, Ozaslan M, Kilic HI. et al. Nephroprotective effect of diffractaic acid on carbon tetrachloride-induced kidney damage in rats. Biotechnol Biotechnol Equip. 2015; 29(5):1011-6. [DOI:10.1080/13102818.2015.1056754]
10.World Health Organization (WHO). Traditional medicine -growing needs and potential [Internet]. 2002 [Updated 2002] Available from https://apps.who.int/iris/handle/10665/67294
11.Mishra S, Pani SR, Sahoo S. Anti-nephrotoxic activity of some medicinal plants from tribal rich pockets of Odisha. Pharmacognosy Res. 2014; 6(3):210-7. [DOI:10.4103/0974-8490.132598] [PMID] [PMCID]
12.Ojha S, Venkataraman B, Kurdi A, Mahgoub E, Sadek B, Rajesh M. Plant-Derived Agents for Counteracting Cisplatin-Induced Nephrotoxicity. Oxid Med Cell Longev. 2016; 2016:4320374. [DOI:10.1155/2016/4320374] [PMID] [PMCID]
13.Jedage HD, Manjunath KP. Phytochemical, pharmacological evaluation of Morinda pubescens J.E.Sm. bark extract for nephroprotective activity. Ayu. 2016; 37(3-4):244-9. [DOI:10.4103/ayu.AYU_196_15] [PMID] [PMCID]
14.Nyarko RA, Larbie C, Anning AK, Baidoo PK, Emikpe BO, Oyagbemi AA, et al. Griffonia simplicifolia (DC.) Baill. attenuates gentamicin and cisplatin-induced nephrotoxicty in rats. Comp Clin Pathol. 2019; 28:1293-304. [DOI:10.1007/s00580-019-02934-x]
15.Mallik J, Das P, Das S. Pharmacological activity of Cucumis sativus L. - A complete overview. Asian J Pharm Clin Res Dev. 2013; 1(1):1-6. http://ajprd.com/index.php/journal/article/view/1
16.Larbie C, Mills-Robertson FC, Dowuona JNN, Tofeh JJ, Torkornoo D, Asare CY. Phytochemical constituents and biological activity of fractions of stem and leaves of Acalypha inferno. J Pharm Res Int. 2017; 20(4):1-12. [DOI:10.9734/JPRI/2017/38675]
17.Larbie C, Nyarko HN, Tofah JJ, Torkornoo D. Aqueous ethanolic extract of Acalypha inferno leaves is safe in animal. Aqueous ethanolic extract of Acalypha inferno leaves is safe in animals. Int J Pharm Sci Res. 2018a; 9(6):2273-80. [DOI:10.13040/IJPSR.0975-8232.9(6).2273-80]
18.Larbie C, Oduro J, Tofah JJ. Aqueous ethanolic extract of Acalypha inferno accelerates the clearance of glucose in normoglyaecemic rats. J Pharm Innov. 2018b; 7(2):79-83. http://www.thepharmajournal.com/archives/?year=2018&vol=7&issue=2&ArticleId=1698
19.Larbie C, Emikpe BO, Oyagbemi AA, Nyarko R, Jarikre T, Adjei CO, et al. Acalypha wilkesiana ‘Inferno’ hydroethanolic leaves extract has protective effect on CCl4-induced subacute toxicity in animals. Biomed Res Ther. 2020; 7(5):3778-88. [DOI:10.15419/bmrat.v7i5.605]
20.Putri ANM, Hakim RW, Purwaningsih EH, Krisnamurti DGB. Kidney protective effect of Acalypha indica linn. root extract in high-fructose and high cholesterol diet-fed rats. J Phys Conf Ser. 2019; 1246:012041. [DOI:10.1088/1742-6596/1246/1/012041]
21.Anokwuru CP, Anyasor GN, Shokunbi OS, Sopekan B, Osinuga OK, Afolabi OE, et al. Chemoprotective activity of aqueous leaf extract of Acalypha wilkesiana against cyclophosphamide-induced toxicity in rats. Asian Pac J Trop Med. 2019; 12(9):409-15. [DOI:10.4103/1995-7645.267584]
22.National Research Council. Guide for care and use of laboratory animal, 8th edition [Internet]. 2011. Available from: https://www.ncbi.nlm.nih.gov/books/NBK54050/
23.Chatterjee P, Mukherjee A, Nandy S. Protective effects of the aqueous leaf extract of Aloe barbadensis on gentamicin and cisplatin-induced nephrotoxic rats. Asian Pac J Trop Biomed. 2012; 2(3):S1754-63. [DOI:10.1016/S2221-1691(12)60490-0]
24.Wokes F, Still BM. The estimation of protein by the biuret and Greenberg methods. Biochem J. 1942; 36(10-12):797-806. [DOI:10.1042/bj0360797] [PMID] [PMCID]
25.Oyagbemi AA, Omobowale TO, Akinrinde AS, Saba AB, Ogunpolu BS, Daramola O. Lack of reversal of oxidative damage in renal tissues of lead acetate-treated rats.Environ Toxicol. 2015; 30(11):1235-43. [DOI:10.1002/tox.21994] [PMID]
26.Varshney R, Kale RK. Effect of calmodulin antagonists on radiation induced lipid peroxidation in microsomes. Int J Radiat Biol. 1990; 58(5):733-43. [DOI:10.1080/09553009014552121] [PMID]
27.Jollow DJ, Mitchell JR, Zampaglione N, Gillette JR. Bromobenzene-induced liver necrosis; protective role of glutathione and evidence for 3, 4 bromobenzene oxide as the hepatotoxic metabolite. Pharmacology. 1974; 11(3):151-69. [DOI:10.1159/000136485] [PMID]
28.Beutler E, Duron O, Kelly BM. Improved method for the determination of blood glutathione. J Lab Clin Med. 1963; 61:882-8. [PMID]
29.Habig WH, Pabst MJ, Jakoby WB. Glutathione S-Transferases: the first enzymatic step in mercapturic acid formation. J Biol Chem. 1974; 249(22):7130-9. [PMID]
30.Adewuyi A. Otuechere CA, Adebayo OL, Anazodo C, Pereira FV. Renal toxicological evaluations of sulphonated nanocellulose from Khaya sengalensis seed in Wistar rats. Chem Biol Interact. 2018; 284:56-68. [DOI:10.1016/j.cbi.2018.02.015] [PMID]
31.Jarikre TA, Emikpe BO. First report of immunohistochemical detection of Pestedes petit ruminants, parainfluenza 3 and respiratory syncytial viral antigens in lungs of Nigerian goats. J Immunoassay Immunochem. 2017;38(5):555-568. [DOI:10.1080/15321819.2017.1349669] [PMID]
32.Nasri H. Renal cell protection of erythropoietin beyond correcting the anemia in chronic kidney disease patients. Cell J. 2014; 15(4):378-80. [PMID] [PMCID]
33.Shastry I, Belurkar S. The spectrum of red blood cell parameters in chronic kidney disease: A study of 300 cases. J Appl Hematol. 2019; 10(2):61-6. [DOI:10.4103/joah.joah_13_19]
34.Shelkea TT, Bhaskarb VH, Adkara PP, Jhaa U, Oswala J. Nephroprotective activity of ethanolic extract of stem bark of Crateava nurvala Buch Hum. Int J Pharm Sci Res. 2011; 2(10):2712-7. [DOI:10.13040/IJPSR.0975-8232.2(10).2712-17]
35.Kinsey GR, Li L, Okusa MD. Inflammation in acute kidney injury. Nephron Exp Nephrol. 2008; 109(4):e102-7. [DOI:10.1159/000142934] [PMID] [PMCID]
36.Turini ME, DuBois RN. Cyclooxygenase-2: A therapeutic target. Annu Rev Med. 2002; 53:35-57. [DOI:10.1146/annurev.med.53.082901.103952] [PMID]
37.Cortvrindt C, Speeckaert R, Moerman A, Delanghe JR, Speeckaert MM. The role of interleukin-17A in the pathogenesis of kidney diseases. Pathology. 2017; 49(3): 247-58. [DOI:10.1016/j.pathol.2017.01.003] [PMID]
38.Lavoz C, Matus YS, Orejudo M, Carpio JD, Droguett A, Egido J, et al. Interleukin-17A blockade reduces albuminuria and kidney injury in an accelerated model of diabetic nephropathy. Kidney Int. 2019; 95(6):1418-32. [DOI:10.1016/j.kint.2018.12.031] [PMID]
39.Duvallet E, Semerano L, Assier E, Falgarone G, Boissier MC. Interleukin-23: A key cytokine in inflammatory diseases. Ann Med. 2011; 43(7):503-11. [DOI:10.3109/07853890.2011.577093] [PMID]
40.Vukelic M, Laloo A, Kyttaris V. Elevated serum interleukin-23 level in patients with systemic lupus erythematosus is associated with disease activity, clinical and immunological markers [abstract]. Arthritis Rheumatol. 2019; 71(suppl 10). https://bit.ly/33uGrXh
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |





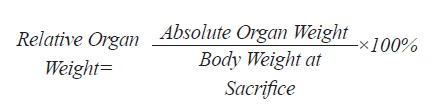
.png)