Introduction
iabetes Mellitus (DM) is a heterogeneous group of metabolic disorders, affecting the lipid, carbohydrate and protein metabolism. It is characterized by hyperglycemia and deficiencies in insulin secretion, and/or insulin resistance [1]. Diabetes is believed to be the most prevalent cause of morbidity and mortality in low to middle-income countries than in rich nations [2]. It is projected that the global incidence of diabetes is escalating yearly, thereby leading to a major burden on the health sector, mainly in developing countries, with the urban population having the highest prevalence [3]. Recent data indicate that about 150 million people worldwide are diabetic, which is expected to double by the year 2025 [4, 5].
The complications arising from diabetes correlate with persistently elevated serum glucose levels, arising from free radicals’ production and leading to oxidative stress in tissues [6, 7]. In this context, antioxidants have been effectively employed to lower the severity of DM complications. The effect of diabetes, like most chronic diseases, on both the individuals and society are extensive in terms of personal income, general productivity and burden on the healthcare system [8].
Effective treatment for diabetes is mainly through the use of oral hypoglycemics, such as thiazolidines and sulfonylureas; however, they have side effects, such as hypoglycemia and atherogenesis [9]. That is why efforts are made to search for alternative treatments, such as herbal medicines with low cost and side effects. This has prompted studies, investigating herbal extracts and their efficacies in the management of DM. These efforts have led to the identification of various plant extracts with hypoglycemic effects in animal models and humans [10].
Reducing postprandial blood glucose levels through the inhibition of carbohydrate hydrolyzing enzymes has been one of the therapeutic strategies toward the management of diabetes [11]. Hydrolysis of dietary carbohydrate, or starch, is a major source of blood glucose in humans. This breakdown process occurs through the actions of α-glucosidase and α-amylase in the intestine. Thus, inhibiting these enzymes can drastically lower the postprandial surge of blood glucose, and provide for an effective management of type II diabetes [12].
Manihot esculenta Crantz (M. esculanta) known as Cassava in Nigeria, is a tropical perennial plant of the Euphorbiaceae family. Cassava is a major staple in most tropical countries, including Nigeria. The roots of this plant are a major source of carbohydrates, while the leaves are consumed as vegetables. They are rich in vitamins A and C, proteins and minerals, particularly Mg, Zn, Fe and Mn [13, 14]. Also, the leaves are rich in phytochemicals, which are valuable as natural antioxidants, and their consumption has been linked to reducing the risks of cancer, hypertension, diabetes, obesity and heart diseases [14, 15]. The leaves also contain some toxins, such as cyanides; therefore, caution must be exercised in their consumption [16, 17]. The toxicity issue affects the nutritive value and acceptability of the leaves, which could be lowered by drying, boiling and pounding for long periods. It has been stated earlier that when the leaves of cassava are pounded or allowed to stand for a period of 5 hour under shade or when washed thoroughly with water three times, between 72% and 99% of the cyanide content could be reduced [13, 18].
Medicinally, the leaves extract is used in the management of various disorders, such as rheumatism, fever, diarrhea, headache and loss of appetite. The anti-inflammatory and antimicrobial properties, and the analgesic and anthelmintic effects of Cassava leaves have been reported previously [13, 19-21]. However, there are no reports available on the effects of the Cassava leaves extract on α-glucosidase and α-amylase. The aim of this study was to evaluate the in vitro inhibitory effects of the two extracts derived from Cassava leaves on α-glucosidase and α-amylase, and the total phenolic contents.
Material and Methods
Chemicals & Reagents: Foline Ciocalteu (FC) reagent, Gallic acid, sodium carbonate anhydrous, sodium nitrite, aluminum chloride anhydrous, sodium hydroxide, 3,5 Dinitro Salicylic Acid (DNSA), porcine pancreatic α-amylase and yeast α-glucosidase and catechin were purchased from Sigma-Aldrich (St. Louise, USA). Other chemicals were of analytical grades and obtained from Merck (Darmstadt, Germany).
Plant Collection & Processing: Fresh leaves of M. esculenta were collected from site III of the Delta State University and their authenticity were confirmed at the Herbarium of Plant Science Department, University of Benin, Benin City, Nigeria (Voucher # UBHM372). The leaves were thoroughly washed three times with water, air-dried at room temperature for ten days, and were powdered. The leaves (50 g) were extracted in 200 ml of ethanol at room temperature for 24hr. This process was repeated for another 24 h and the extract was concentrated at 40°C under vacuum to dryness and was refrigerated until further use. The extraction with acetone was performed similarly to the procedure used for the ethanol and the material was refrigerated similarly. The dried extracts were dissolved in Dimethyl Sulfoxide (DMSO) to obtain the concentrations required for the subsequent experiments.
α-Glucosidase Inhibition Assay: The α-glucosidase inhibitory effect of M. esculenta leaf extracts was achieved following the procedure of Kwon et al. [22] and as described by Okoro et al. [23]. Acarbose was used in this assay as the positive control. The results are stated in percentage as derived from the equation shown below, under 𝛼-amylase inhibition assay.
α-Amylase Inhibition Assay: This test was made by the modified method of McCue et al. [24] and as described by Okoro et al. [23]. Acarbose was used in this assay as the positive control. The α-amylase inhibitory effect was calculated using the following equation: % Inhibition=[(Ac - Ae) /Ac] x 100; where: Ac=absorbance of the control and Ae=sample absorbance.
Total Phenolic Content: The total phenolic content of the M. esculenta leaf extract was evaluated spectrophotometrically, using the Folin-Ciocalteu’s reagent as described by Singleton et al. [25]. Then, the total phenol content was calculated as the Gallic acid equivalent.
Total Flavonoid Content (TFC) The total flavonoid contents of both extracts were evaluated by the aluminum chloride colorimetric method of Chang, et al. [26]. The TFC was determined based on a calibration curve, in which quercetin was used in the calibration curve and the TFC was expressed as mg of quercetin equivalent per gram M. esculenta (mg QCE/g).
Statistical Analysis: The data were statistically analyzed by One-way Analysis of Variance (ANOVA) followed by Sidak’s test in Graphpad Prism, version 6.0 (Graph Pad Software, San Diego, CA, USA), expressed as Mean±Standard Deviation (SD) at a confidence limits of 95% (P≤0.05).
Results
α-Glucosidase Inhibitory Studies: The results of α-glucosidase inhibitory effects are shown in Figure 1. The results revealed a concentration dependent increase as percent inhibition for both acetone and ethanol extracts versus the positive control drug, Acarbose. The α-glucosidase inhibitory activity, expressed as a function of the extract concentration, displayed the inhibition ranging from 13.57±1.00 to 78.83±4.52 percent at 0.25 and 2.00 mg/mL, respectively, for the ethanol extract. Also, the percent inhibition by the acetone extract was from 6.49±1.27 to 70.77±6.87 percent at 0.25 and 2.00 mg/mL, respectively. The reference drug, Acarbose, demonstrated inhibition values of 7.60±0.77 and 64.56±3.63 at 0.25 and 2.00 mg/mL, respectively. Consequently, the highest percent inhibition was noted for the ethanol extract. This was further demonstrated by the lower IC50 value (0.77±0.03 mg/mL), while the acetone extract resulted in a minimal inhibition with the IC50 value of 1.46±0.17 mg/mL (Table 1). The extracts and positive control displayed percent inhibition in the following decreasing order: ethanol extract > Acarbose > acetone extract. Thus, the ethanol extract displayed a significantly higher inhibition percent than the acetone extract (P<0.05).

α-Amylase Inhibitory Studies: The results of α-amylase inhibitory effects are shown in Figure 2. The percent α-amylase inhibition results, expressed as a function of extract concentration, shows the inhibition trend of both extracts of Cassava leaves and Acarbose (standard). The percent inhibition ranged from 10.80±0.74 to 65.81±2.53 at 0.25 and 2.00 mg/mL for the ethanol extract, respectively. The percent inhibition for the acetone extract ranged from 6.42±1.11 to 55.93±1.85 at 0.25 and 2.00 mg/mL, respectively. Similarly, the standard drug displayed inhibition values of 9.70±1.31 and 86.07±2.14 at 0.25 and 2.00 mg/mL, respectively. Thus, the highest percent inhibition was shown by the ethanol extract (IC50=1.29±0.02 mg/mL) and the least inhibition was recorded for the acetone extract (IC50=1.66±0.05 mg/mL), as represented by the data in Table 1. Although, the positive control drug displayed the highest inhibition percent overall, the ethanol extract was more effective than the other extract, as it displayed a significantly higher percent inhibition of α-amylase than the acetone extract did (P<0.05).

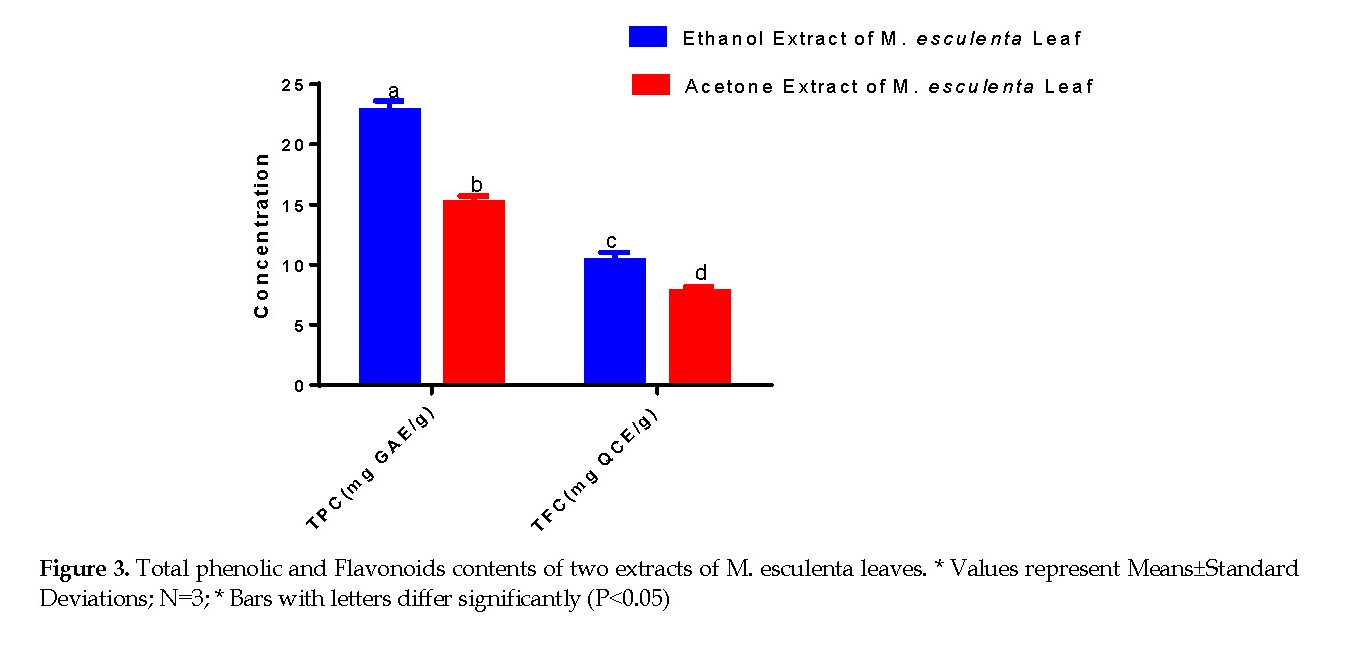
Total Phenolic and Flavonoid Contents: Figure 3 presents the Total Phenolic Content (TPC) of the two extracts of M. esculenta leaves, expressed as mg Gallic acid equivalents per grams of the extract. The crude ethanol extract of M. esculenta leaves contained 22.81±0.82 mg Gallic acid equivalent per gram of the extract. The acetone extract produced 15.13±0.58 mg Gallic acid equivalent per gram of extract. Thus, both extracts displayed varying contents of total phenol with the ethanol extract producing a significantly higher content than did the acetone extract (P<0.05). Similarly, the influence of the solvents on the Total Flavonoid Content (TFC) was comparable to that of the TPC. The maximum TFC of 10.36±0.64 expressed as the quercetin equivalent (mg QCE/g) was found for the ethanol extract, while lower (7.71±0.45) content was observed for the acetone extract. The ethanol extract exhibited the highest contents of both phenols and flavonoids between the two extracts.
Discussions
Diabetes mellitus is a metabolic disease characterized by hyperglycemia. A carbohydrate-rich diet usually leads to a sharp rise in blood glucose level, because the complex carbohydrates from food are rapidly absorbed, aided by α-amylase and α-glucosidase enzymes in the intestine. These enzymes break down carbohydrates into simple, absorbable monosaccharides. By inhibiting these enzymes, the absorption of glucose is delayed, and the surge in blood glucose is precluded, thus making this a different therapeutic approach in the management of diabetic patients [22].
The regularly used synthetic drugs, such as Miglitol and Acarbose have been broadly used in the management of DM as standard, clinical drugs. They act by inhibiting α-amylase and
α-glucosidase, thus lowering the blood glucose level. However, they have adverse effects, such as liver toxicity, flatulence and diarrhea [11]. Hence, using plant-based inhibitors of α-glucosidase may be a promising therapeutic approach for the treatment of patients with type II diabetes [27]. This study was undertaken to evaluate the in vitro α-glucosidase and α-amylase inhibitory effects, plus their total phenolic and flavonoid contents.
The inhibitory effect of α-amylase of the two extracts is likely to be due to the flavonoid and phenolic contents. The polyphenols have been reported to reduce oxidative stress and inhibit carbohydrate hydrolyzing enzymes by binding to proteins [28]. Flavonoids are polyphenolic compounds that have been described to have both α-glucosidase and α-amylase inhibitory activity [29, 30]. The inhibitory effects of flavonoids have been linked to interactions that obstruct the reaction between starch and α-amylase, thus inhibiting the digestion of starch [31]. The results of this study are in agreement with earlier reports that suggested a positive association between the flavonoid content, total polyphenols, and the ability to inhibit α-amylase and α-glucosidase [29, 32].

The findings of this study confirmed the inhibitory effects of the extracts on α-glucosidase. This study also demonstrated a trend similar to that of α-amylase where the ethanol extract gave the lowest IC50 value, suggesting a higher inhibitory activity of the extract against the enzymes compared to those for the acetone extract. Overall, both extracts demonstrated strong inhibitory activities similar to Acarbose. Thus, it may be inferred that the extracts, especially the ethanolic one, would be a promising plant source of hypoglycemic agent with lower side effects than Acarbose for the management of diabetic patients.
A review of literature revealed that the leaves of M. esculenta plant are rich in terpenes, flavonoids, tannins, alkaloids, saponins and phenols, all of which are believed to be effective inhibitors of α-glucosidase [33]. This study found a weak α-amylase and a considerable α-glucosidase inhibitory activity by both extracts tested. An obvious association between polyphenols and the anti-diabetic effects of herbal extracts has previously been reported [34]. Similarly, studies have revealed that the antioxidant properties of plants are directly proportional to their phenolic contents. The α-glucosidase and α-amylase inhibitory effects of flavonoids and tannins have also been demonstrated [35]. In the current study, both extracts of M. esculenta leaves displayed varying contents of total phenolic agents with the ethanol extract, having a significantly higher phenolic content than the acetone extract. This finding is not consistent with an earlier report by Suresh, et al. [21], where a higher total phenolic content was found in the acetone extract of M. esculenta leaves than in the ethanolic extract, but was consistent with those reported by Khan, et al. [36].
Phenolic compounds are known secondary metabolites which act as antioxidants as a result of their ability to release hydrogen, quench singlet oxygen, and act as chelators of metal ions [37, 38]. Oxidative stress plays a major role in the development of diabetic complications, and free radicals are known to be generated in diabetes due to glucose oxidation and the successive oxidative breakdown of glycosilated proteins [39].
Flavonoids are the secondary metabolites with antioxidant activity, and their potency depends upon the number and position of the free OH groups [40, 41]. It is suggested that genetic diversity, environmental, biological, and seasonal differences largely affect the flavonoid contents in plants [42]. Flavonoids are a group of polyphenolic compounds with a benzo-γ-pyrone structure, and are beneficial in various ways to the plants [43]. The usefulness of flavonoids as antimicrobial, anti-angiogenic, antiulcer, anti-arthritic and anticancer agents have been reported [44]. The preventive roles of beverages and phenolic-rich foods against diseases, like cancer, inflammation, heart disease, neurodegenerative diseases, arthritis, immune related diseases, and diabetes have also been confirmed [45]. In this study, the presence of flavonoid and phenolic compounds in the extracts of M. esculenta leaves correlated with the health values of the plant, particularly their anti-hyperglycemic effects.
Limitations of the Study: The isolation of active components responsible for the observed inhibitory activities of the extracts against α-glucosidase and α-amylase, and its structural elucidation that might be useful to developing new anti-diabetic agents were not carried out. This was because the study was aimed at an in-vitro assessment of α-glucosidase and α-amylase inhibitory activity of the ethanol and acetone extracts of M. esculenta.
Recommendations for Future Research: Based on our results, we recommend that further research on the extracts be undertaken to elucidate the molecular components responsible for the observed inhibition of the tested enzymes.
Conclusion
The in vitro α-glucosidase and α-amylase inhibitory effects of both extracts of M. esculenta leaves, as investigated in this study, indicated that they possessed significant inhibitory activities against both enzymes dose dependently, with the ethanol extract demonstrating the most effective inhibition of α-glucosidase and α-amylase. The inhibitory effects of the extracts were also positively related with their total flavonoid and phenolic contents. Thus, the extracts have anti-hyperglycemic properties and are potential candidates to be used in the management of diabetes and the resultant oxidative stress.
Ethical Considerations
Compliance with ethical guidelines
The technical support provided by Mrs. Edith Omozefe Okoro of Emmanuel Medical Laboratory in Abraka, Nigeria, is greatly appreciated. This study did not use animals or human subjects; however, the Ehical principles in the use of chemicals and plants were followed as set by Delta State University, Abraka, Nigeria.
Funding
The author funded this study and did not receive any financial support from other internal or external sources.
References
Huang DW, Shen SC. Caffeic acid and cinnamic acid ameliorate glucose metabolism via modulating glycogenesis and gluconeogenesis in insulin-resistant mouse hepatocytes. J Funct Foods. 2012; 4(1):358-66. [DOI:10.1016/j.jff.2012.01.005]
Georg P, Ludvik B. Lipids and diabetes. J Clin Basic Cardiol. 2000; 3(3):159-62. https://www.kup.at/kup/pdf/116.pdf
World Health Organization. Diabetes [Internet]. 2020 [Updated 2020 June 8]. Available from: Available at: https://www.who.int/news-room/fact-sheets/detail/diabetes
World Health Organization. Diabetes, Geneva: World Health Organization, 2018. Available at: https://www.who.int/news-room/fact-sheets/detail/diabetes.
Field MJ, Pollock C, Harris D. The renal system: Systems of the body series. 2nd ed. London: Churchill Livingstone; 2011. https://books.google.com/books?id=cYfGAAAAQBAJ&dq
Asmat U, Abad Kh, Ismail Kh. Diabetes mellitus and oxidative stress-a concise review. Saudi Pharm J. 2016; 24(5):547-53. [DOI:10.1016/j.jsps.2015.03.013] [PMID] [PMCID]
Tiwari BK, Pandey KB, Abidi AB, Rizvi SI. Markers of oxidative stress during diabetes mellitus. J Biomark. 2013; 2013:378790. [DOI:10.1155/2013/378790] [PMID] [PMCID]
Yen ST, Tan AKG, Mustapha FI. Awareness of diabetes, hypertension, and hypercholesterolemia in Malaysia. J Diabetes. 2017; 9(9):874-83. [DOI:10.1111/1753-0407.12502] [PMID]
Hussein Z, Taher SW, Gilcharan Singh HK, Chee Siew Swee W. Diabetes care in Malaysia: Problems, new models, and solutions. Ann Glob Health. 2015; 81(6):851-62. [DOI:10.1016/j.aogh.2015.12.016] [PMID]
Modak M, Dixit P, Londhe J, Ghaskadbi S, Devasagayam TPA. Indian herbs and herbal drugs used for the treatment of diabetes. J Clin Biochem Nutr. 2007; 40(3):163-73. [DOI:10.3164/jcbn.40.163] [PMID] [PMCID]
Nair SS, Kavrekar V, Mishra A. In vitro studies on alpha amylase and alpha glucosidase inhibitory activities of selected plant extracts. Eur J Exp Biol. 2013; 3(1):128-32. https://www.imedpub.com/articles/in-vitro-studies-on-alpha-amylase-and-alpha-glucosidase-inhibitory-activities-of-selected-plant-extracts.php?aid=11467
Kwon YI, Apostolidis E, Shetty K. Inhibitory potential of wine and tea against α‐amylase and α‐glucosidase for management of hyperglycemia linked to type II diabetes. J Food Biochem. 2008; 32(1):15-31. [DOI:10.1111/j.1745-4514.2007.00165.x]
Montagnac JA, Davis CR, Tanumihardjo SA. Processing techniques to reduce toxicity and antinutrients of cassava for use as a staple food. Compr Rev Food Sci Food Saf. 2009; 8(1):17-27. [DOI:10.1111/j.1541-4337.2008.00064.x]
Fasuyi AO. Nutrient composition and processing effects on cassava leaf (Manihot esculenta, Crantz) antinutrient. Pak J Nutr. 2005; 4(1):37-42. [DOI:10.3923/pjn.2005.37.42]
Asif M. The role of fruits, vegetables, and spices in diabetes. Int J Nutr Pharmacol Neurol Dis. 2011; 1(1):27-35. [DOI:10.4103/2231-0738.77527]
Okoro IO. Effects of extraction solvents on the antioxidant and phytochemical activities of Manihot esculenta leaves. Iran J Toxicol. 2020; 14(1):51-8. http://ijt.arakmu.ac.ir/article-1-774-en.html
Cereda MP, Mattos MCY. Linamarin - the toxic compound of cassava. J Venom Anim Toxins. 1996; 2(1):6-12. [DOI:10.1590/S0104-79301996000100002]
Howard Bradbury J, Denton IC. Mild method for removal of cyanogens from cassava leaves with retention of vitamins and protein. Food Chem. 2014; 158:417-20. [DOI:10.1016/j.foodchem.2014.02.132] [PMID]
Adeyemi OO, Yemitan OK, Afolabi L. Inhibition of chemically induced inflammation and pain by orally and topically administered leaf extract of Manihot esculenta Crantz in rodents. J Ethnopharmacol. 2008; 119(1):6-11. [DOI:10.1016/j.jep.2008.05.019]
Haj Hashemi VA, Ghanadi AR, Mosavi D. [Analgesic and antiinflammatory effects of total extract, flavonoid fraction and volatile oil of Salvia hydrangea (Persian)]. J Res Med Sci. 2000; 5(Suppl 2):10-14. https://www.sid.ir/fa/journal/ViewPaper.aspx?ID=32932
Suresh R, Saravanakumar M, Suganyadevi P. Anthocyanins from Indian cassava (Manihot esculenta Crantz) and its antioxidant properties. Int J Pharm Sci Res. 2011; 2(7):1819-28. https://ijpsr.com/bft-article/anthocyanins-from-indian-cassava-manihot-esculenta-crantz-and-its-antioxidant-properties/
Kwon YI, Vattem DA, Shetty K. Clonal herbs of Lamiaceae species against diabetes and hypertension. Asia Pac J Clin Nutr. 2006; 15:424-32.
Okoro IO, Umar IA, Atawodi SE, Anigo KM. In vitro and in vivo antihyperglycemic effect of active fraction of Cleome rutidosperma DC. Int J Pharm Pharm Sci. 2015; 7(1):289-95. https://innovareacademics.in/journals/index.php/ijpps/article/view/3337
McCue P, Vattem D, Shetty K. Inhibitory effect of clonal oregano extracts against porcine pancreatic amylase in vitro. Asia Pac J Clin Nutr. 2004; 13(4):401-8. [PMID]
Singleton VL, Orthofer R, Lamuela-Raventós RM. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. In: Packer L. Oxidants and Antioxidants Part A, Methods in Enzymology. Vol. 299. Cambridge: Academic Press; 1999. pp. 152-78. [DOI:10.1016/S0076-6879(99)99017-1]
Chang CC, Yang MH, Wen HM, Chern JC. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J Food Drug Anal. 2002; 10(3):178-82. https://cutt.ly/Eo2M8aO
Mcdongall GJ, Stewart D. The inhibitory effects of berry polyphenols on digestive enzymes. BioFactors. 2005; 23(4):189-95. [DOI:10.1002/biof.5520230403] [PMID]
Bothon FTD, Debiton E, Avlessi F, Forestier C, Teulade JC, Sohounhloue DKC. In vitro biological effects of two anti-diabetic medicinal plants used in Benin as folk medicine. BMC Complement Altern Med. 2013; 13:51. [DOI:10.1186/1472-6882-13-51] [PMID] [PMCID]
Li K, Yao F, Xue O, Fan H, Yang L, Li X, et al. Inhibitory effects against α-glucosidase and α-amylase of the flavonoids-rich extract from Scutellaria baicalensis shoots and interpretation of structure-activity relationship of its eight flavonoids by a refined assign-score method. Chem Cent J. 2018; 12(1):82. [DOI:10.1186/s13065-018-0445-y] [PMID] [PMCID]
Kalita D, Holm DG, LaBarbera DV, Petrash JM, Jayanty SS. Inhibition of α-glucosidase, α-amylase, and aldose reductase by potato polyphenolic compounds. PLoS One. 2018; 13(1):e0191025. [DOI:10.1371/journal.pone.0191025] [PMID] [PMCID]
Gu C, Zhang H, Yusolf Putri C, Ng K. Evaluation of α-amylase and α-glucosidase inhibitory activity of flavonoids. Int J Food Nutr Sci. 2015; 2(2):174-9. [DOI:10.15436/2377-0619.15.042]
Mai TT, Thu NN, Tien PG, Chuyen NV. Alpha-glucosidase inhibitory and antioxidant activities of Vietnamese edible plants and their relationships with polyphenol contents. J Nutr Sci Vitaminol (Tokyo). 2007; 53(3):267-76. [DOI:10.3177/jnsv.53.267] [PMID]
Yin Z, Zhang W, Feng F, Zhang Y, Kang W. α-Glucosidase inhibitors isolated from medicinal plants. Food Sci Hum Wellness. 2014; 3(3-4):136-74. [DOI:10.1016/j.fshw.2014.11.003]
Maria John KM, Rajesh J, Mandal AKA, Natarajan S. Antioxidant and antimicrobial activity of individual catechin molecules: A comparative study between gallated and epimerized catechin molecules. Eur J Exp Biol. 2011; 1(3):145-53. https://www.imedpub.com/articles/antioxidant-and-antimicrobial-activity-of-individual-catechin-moleculesa-comparative-study-between-gallated-and-epimerized-catechi.pdf
Poongunran J, Perera HKI, Fernando WIT, Jayasinghe L, Sivakanesan R. α-Glucosidase and α-amylase inhibitory activities of nine Sri Lankan antidiabetic plants. Br J Pharm Res. 2015; 7(5):365-74. [DOI:10.9734/BJPR/2015/18645]
Khan MS, Yusufzai SK, Kimin L, Nabila Binti Jabi NA. Determination of chemical composition, total flavonoid content, total phenolic content and antioxidant capacity of various crude extracts of Manihot esculenta crantz leaves. Int J Res Appl Sci Eng Technol. 2018; 6(4):2433-43. [DOI:10.22214/ijraset.2018.4413]
Moein S, Pimoradloo E, Moein MR, Vessal M. Evaluation of antioxidant potentials and α-amylase inhibition of different fractions of labiatae plants extracts: As a model of antidiabetic compounds properties. Biomed Res Int. 2017; 2017:7319504. [DOI:10.1155/2017/7319504] [PMID] [PMCID]
Kasote DM, Katyare SS, Hegde MV, Bae H. Significance of antioxidant potential of plants and its relevance to therapeutic applications. Int J Biol Sci. 2015; 11(8):982-91. [DOI:10.7150/ijbs.12096] [PMID] [PMCID]
Mehta Jl, Rasouli N, Sinha AK, Molavi B. Oxidative stress in diabetes: A mechanistic overview of its effects on atherogenesis and myocardial dysfunction. Int J Biochem Cell Biol. 2006; 38(5-6):794-803. [DOI:10.1016/j.biocel.2005.12.008] [PMID]
Panche AN, Diwan AD, Chandra SR. Flavonoids: An overview. J Nutr Sci. 2016; 5:e47. [DOI:10.1017/jns.2016.41] [PMID] [PMCID]
Kumar Sh, Pandey AK. Chemistry and biological activities of flavonoids: An overview. Sci World J. 2013; 2013:162750. [DOI:10.1155/2013/162750] [PMID] [PMCID]
Kumar V, Roy BK. Population authentication of the traditional medicinal plant Cassia tora L. based on ISSR markers and FTIR analysis. Sci Rep. 2018; 8(1):10714. [DOI:10.1038/s41598-018-29114-1] [PMID] [PMCID]
Praveen Kumar P, Kumaravel S, Lalitha C. Screening of antioxidant activity, total phenolics and GC-MS study of Vitex negundo. Afr J Biochem Res. 2010; 4(7):191-5. https://academicjournals.org/article/article1380110714_Kumar%20et%20al.pdf
John B, Sulaiman CT, George S, Reddy VRK. Total phenolics and flavonoids in selected medicinal plants from Kerala. Int J Pharm Pharm Sci. 2014; 6(1):406-8. https://www.researchgate.net/publication/267031901
Chalise JP, Acharya K, Gurung N, Bhusal RP, Gurung R, Skalko-Basnet N, et al. Antioxidant activity and polyphenol content in edible wild fruits from Nepal. Int J Food Sci Nutr. 2010; 61(4):425-32. [DOI:10.3109/09637481003591590] [PMID]








































