Introduction
The ability of chemical, physical, or biological agents to produce adverse effects on the structural or functional parts of the central and peripheral nervous systems is referred to as neurotoxicity [1]. Neurotoxicity can lead to the disruption or death of neurons, which are the cells responsible for transmitting and processing signals throughout the nervous system. It is the primary contributor to neurodegenerative diseases such as Huntington's, Parkinson's, and Alzheimer's diseases [2]. Neurotoxicity can lead to the onset of neurocognitive impairments, ataxia, incontinence, loss of vision, behavioural problems, sexual dysfunction, etc [3].
Neurotoxicity can occur due to exposure to substances utilised in chemotherapy, medication treatments, and organ transplants, as well as toxic elements, such as mercury and lead, specific food items and food additives, pesticides, etc [4]. Monosodium glutamate (MSG), sodium glutamate, extensively used as a food additive, has been implicated to have neurotoxic effects [5,6].
The MSG, composed of approximately 87.72% glutamate, is recognised as a significant contributor to neurotoxic effects [6,7]. Excessive glutamate in the extracellular space leads to neuronal death in the CNS by excessively stimulating glutamate receptors, a condition known as excitotoxicity [7,8].
The overexcitation of N-methyl-D-aspartate (NMDA) receptors results in an influx of calcium, which stimulates nitric oxide (NO) synthase, glyceraldehyde 3-phosphate dehydrogenase, and cysteine proteases, causing mitochondrial injury and leading to massive energy failure [9,10]. Moreover, the overactivation of the glutamatergic receptors increases the intracellular zinc level, which causes glycolytic dysfunction by interfering with the mitochondrial electron transport chain, inhibiting the citric acid cycle, and increasing the levels of reactive oxygen species [10].
These procedures ultimately result in the death of neurons. Reports suggest that high levels of MSG consumption can result in cognitive decline by increasing the levels of acetylcholinesterase and decreasing dopamine levels [11,12]. Unfortunately, there is currently an unmet need to mitigate the neurotoxic side effects of MSG; therefore, finding an effective treatment that can be integrated into a diet to abrogate this effect is crucial.
Camel milk (CM) is colloquially regarded as the 'white gold of the desert' due to its rich nutritional profile [13]. It is a highly nutritional milk rich in lactoferrins, lysozymes, minerals, proteins, vitamins, and immunoglobulins with lower fat and lactose content [14]. Furthermore, various studies have associated CM with the possible treatment of some pathophysiological disorders, including diabetes, autism, cancer, dropsy, asthma, anaemia, infections, and colitis [15,16]. However, the application of CM to abolish neurotoxicity is predominantly uninvestigated. Therefore, the present study explores the potential therapeutic impact of CM on neurotoxicity triggered by MSG.
Materials and Methods
Drugs and chemicals
The MSG used in this research was obtained from Sigma Chemical in St. Louis, MO, USA. The CM was procured from the Camel Research Institute, King Faisal (Al-Ahsa, Saudi Arabia). All chemicals and drugs used for this study were obtained from reputable companies and were of the highest analytical-grade standards.
Animals
A total of 24 male Wistar rats, weighing 185 and 205 grams, were obtained from an accredited farm house in Nigeria. The rats were then kept in standard cages that provided good environmental conditions. They received a nutritional regimen of standard rat pellets (Tripod Feed Limited, Nigeria) and water ad libitum. The Ethical Review Board of the Physiology Department approved the research protocol issued on 10 January 2024, under the Ethical Approval Number EKSU/P100/2024/01/002. The globally recognised guidelines for the care and use of laboratory animals, established by the Canadian Council on Animal Care and the Guidelines for Protocol Review (NRC, 1997), were strictly followed. The experiment was also in accordance with the guidelines provided by the National Institutes of Health regarding the care and use of laboratory animals.
MSG preparation
In this research, a dosage of 6 g/kg body weight (bw) of MSG was administered [17]. A stock solution was prepared by dissolving 22.0 g of MSG in 1 mL of distilled water. Resulting in a concentration of 600 mg/ml.
Experimental design
Following a two-week acclimatisation period, the animals were randomly assigned to four groups. (n=6 rats each), structured as follows:
Group A (Control): Distilled water (1 ml/kg bw, orally) was administered for 21 days Group B (MSG Control): MSG 6 g/kg bw for 21 days, administered orally
Group C (MSG+CM): Received CM (5 ml/kg bw, orally) 15 minutes after MSG administration Group D (Recovery): MSG was administered for the first 21 days, then left for another 21 days without any administration.
All rats fasted 12 hours overnight were weighed and euthanised using ketamine (40 mg/kg)/xylazine (4 mg/kg) injected intraperitoneally 24 hours after the last administration (day 23). The brain was extracted, weighed, and documented. Post-weighing, the brain was precisely bisected into two equal hemispheres [18]. A portion of the brain tissue was homogenised in a cold phosphate-buffered solution (1:5) using a glass homogeniser, followed by centrifugation at 10,000×g, 4°C for 15 minutes to separate the supernatant from the solution. Both portions were preserved at -20°C for subsequent biochemical assays of oxidative and inflammatory markers. The other brain portions were fixed sufficiently with 10% neutral buffer formalin and preserved at ambient temperature for histopathological evaluation.
Determination of Lipid peroxidation, GSH, antioxidant activities, and Analysis of the Brain Inflammatory Markers
Lipid peroxidation was determined and expressed as the Malondialdehyde (MDA) level using the MDA ELISA kit (Bioassay Tech, China) following the manufacturer's instructions. MDA level was reported in micromoles per gram of tissue (µM/g protein).
The concentrations of Superoxide dismutase (SOD), Glutathione peroxidase (GPx), Glutathione S- S-transferase (GST), and Catalase were assessed using their respective rat ELISA kits (MyBioSource, Inc., US). It was reported in units per milligram of protein (U/mg protein). The glutathione (GSH) concentration was evaluated using the procedure described by Sedlak and Lindsay [19].
The Inflammatory markers (TNFα, IL-1β, Myeloperoxidase, CRP, and NF-kB) were assessed utilising the enzyme-linked immunosorbent assay (ELISA) technique with a standard commercial kit for rats (MyBioSource, Inc., US) according to the producer’s guide. Moreover, NO levels were evaluated utilising a commercial kit which contains Griess reagents (R&D Systems, USA) based on the approach outlined by Griess et al [20]. Determination of the acetylcholine esterase and dopamine levels
The activity of the enzyme Acetylcholine Esterase (AChE) was analysed using rat ELISA kits from Shanghai Sunred Technology Company, following the manufacturer's instructions. Dopamine levels were determined using the ELISA kits supplied by USCN Life Inc., Wuhan, China, according to the producer’s guidelines.
Evaluation of Caspase-3 activity
The eluate formed from the homogenisation and centrifugation of brain samples was utilised for the ELISA technique to measure caspase-3 activity. Following the manufacturer's guidelines, each sample was analysed using a rat caspase-3 ELISA kit from USCN Life Business Co, USA.
Histopathological analysis
Each group's cerebellar tissue sample was separated and accurately preserved in 10% neutral formalin. Then, the tissue was subjected to dehydration using a graduated ethanol series, cleared with xylene, coated in paraffin wax, sliced into 5 μm sections with a microtome, and stained using hematoxylin and eosin. The resulting sections were evaluated by an expert in the field using a light microscope to identify histopathological changes [21].
Statistical analysis
Data analysis was conducted using the GraphPad Prism software (version 9.0, GraphPad Software, Inc.). The findings are presented as mean values along with standard deviation (mean±SD). To compare multiple groups, a one-way analysis of variance (ANOVA) and a Tukey post hoc test were conducted. The threshold for statistical significance was established at p<0.05.
Results
Effect of CM on body weight and brain weight in MSG-exposed rats
The result of the effect of CM on IBW, FBW, BWC, and BrW in MSG-exposed rats is depicted in Table 1. The IBW and FBW showed no significant (p>0.05) difference across all the groups of rats. The BWC showed a notable (p<0.05) decrease and an increase in rats from Groups B and C when compared with control rats, a prominent (p<0.05) increase in Groups C and D rats when compared with Group B, and a significant (p<0.05) decrease in Group D rats when compared with Group C rats. Meanwhile, the BrW showed a significant (p<0.05) decrease in Groups B and C rats compared with Group A rats, and a significant (p<0.05) increase in Groups C and D rats compared with Group B rats.
Table 1. Effect of CM on Body weight and brain weight in rats exposed to MSG
|
Group |
| Group A |
Group B |
Group C |
Group D |
| (Control) |
(MSG-exposed) |
(MSG-exposed+CM) |
(MSG-exposed-R) |
| IBW (g) |
193.00±10.58 |
199.70±7.57 |
196.70±4.73 |
193.00±3.61 |
| FBW (g) |
216.70±11.93 |
210.30±7.64 |
230.30±5.51 |
215.00±4.58 |
| BWC (g) |
23.67±1.53 |
10.67 ±1.53* |
33.67 ±1.53*+ |
22.00±1.00+& |
| BrW (g) |
2.07±0.12 |
1..33±0.06* |
1.63±0.06*+ |
1.87±0.12+ |
Values are mean±SD of three replicates, where *p<0.05 vs control, +p<0.05 vs MSG-exposed, and &p<0.05 vs MSG-exposed+CM, and IBW, FBW, BWC, and BrW were initial body weight, final body weight, body weight change, and brain weight, respectively.
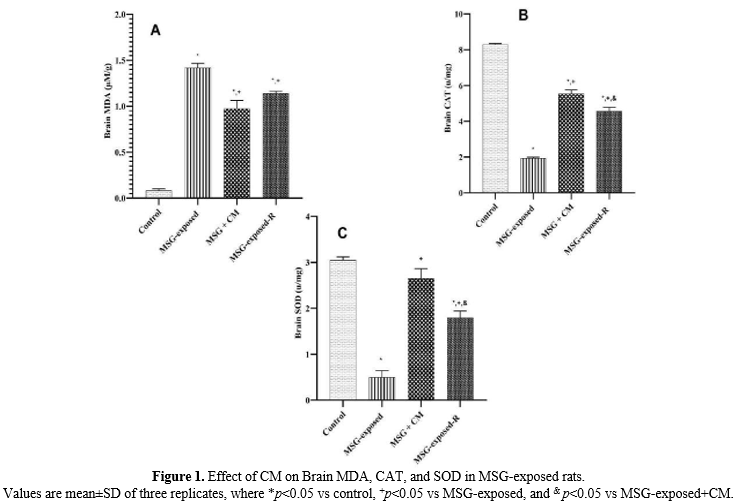
Effect of CM on Brain MDA, CAT, and SOD in MSG-exposed rats
The results of brain MDA, CAT, and SOD are expressed in Figure 1. The results of brain MDA (Figure 1A) showed a significant (p<0.05) increase in all other groups of rats compared with the control rats, and a significant (p<0.05) decrease in Groups C and D rats compared to the Group B rats. However, the brain CAT result (Figure 1B) revealed a notable (p<0.05) decrease in all other groups of rats compared to the control rats, and a significant (p<0.05) increase in the rats of Groups C and D when compared with Group B rats. Similarly, the result of brain SOD (Figure 1C) revealed a significant (p<0.05) decrease in Groups B and D rats when compared with the control rats, and a significant (p<0.05) increase in Groups C and D rats when compared with Group B rats.
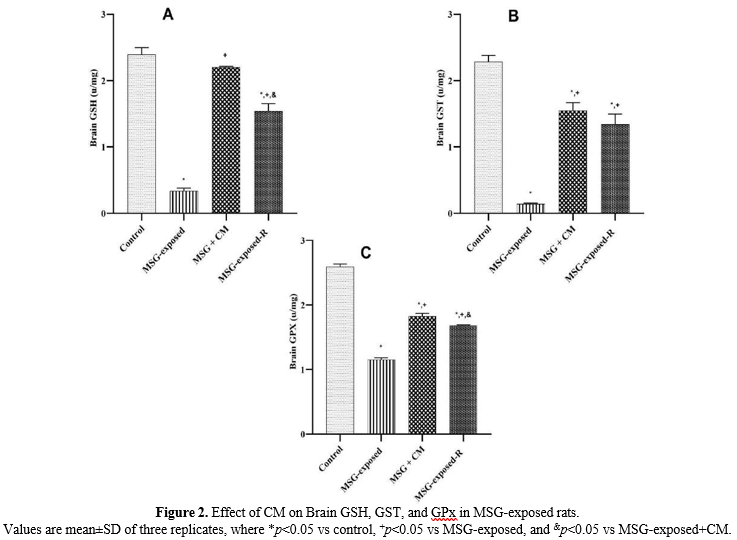
Effect of CM on Brain GSH, GST, and GPx in MSG-exposed rats
The results of brain Glutathione S-transferase (GST), GPx, and GSH were expressed in Figure 2. The results of brain GSH (Figure 2A) showed a significant (p<0.05) decrease in Groups B and D rats compared with the control rats, and a significant (p<0.05) increase in Groups C and D rats compared with Group B rats. Similarly, the result of brain GST and GPx (Figure 2 B and 2 C) revealed a prominent (p<0.05) decrease in all other groups of rats when compared with the control rats, and a significant (p<0.05) increase in Group C and D rats when compared with Group B rats.
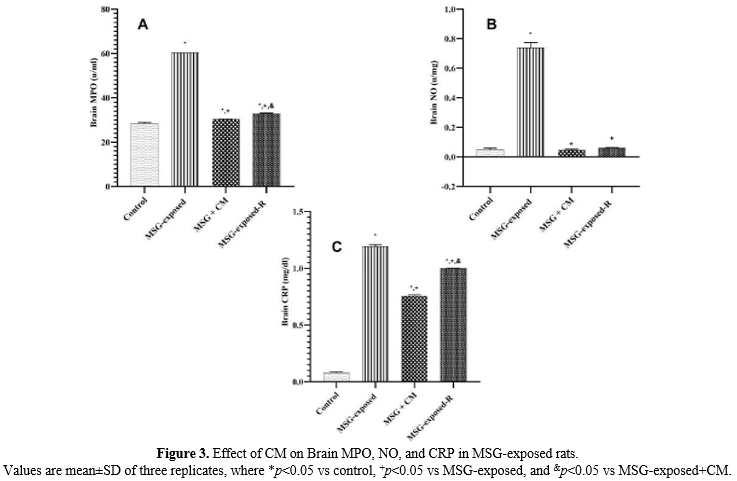
Effect of CM on Brain MPO, NO, and CRP in MSG-exposed rats
The results of Brain MPO, NO, and CRP were expressed in Figure 3. The result of brain MPO and CRP (Figure 3A and Figure 3C) demonstrated a significant (p<0.05) increase in all other groups of rats when compared with the control rats, and a significant (p<0.05) decrease in Groups C and D rats when compared with the Group B rats. Similarly, the result of testicular NO (Figure 3B) revealed a significant (p<0.05) increase in Group B rats only when compared with the control rats, and a significant (p<0.05) decrease in Group C and D rats when compared with Group B rats.
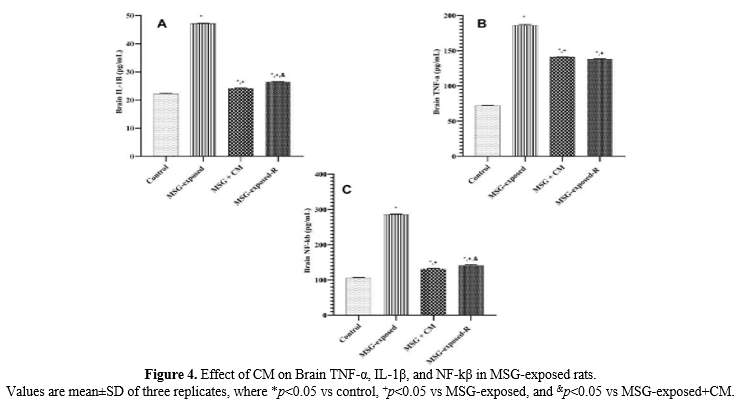
Effect of CM on Brain TNF-α, IL-1β, and NF-kβ in MSG-exposed rats
The findings for brain interleukin-1β (IL-1β), tumor necrosis factor-α, and nuclear factor-kappa B are expressed in Figure 4. The results of brain IL-1β, TNF-α, and NF-kβ (Figure 4 A, B, and C) all demonstrated a significant (p<0.05) increase in all other groups of rats when compared with the control rats, and a significant (p<0.05) decrease in Groups C and D rats when compared with the Group B rats.
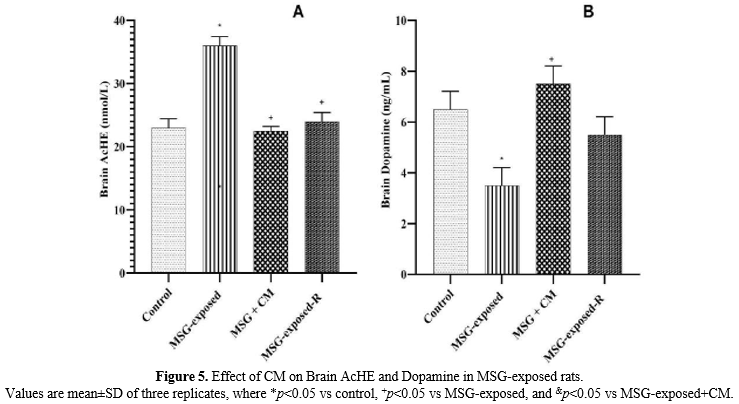
Effect of CM on Brain AcHE and Dopamine in MSG-exposed rats
The results for brain AcHE and dopamine are indicated in Figure 5. The result of AcHE (Figure 5A) revealed a significant (p<0.05) increase in Group B rats when compared with the control rats, and a prominent (p<0.05) decrease in Groups C and D rats when compared with Group B rats. The result of dopamine (Figure 5B) showed a significant (p<0.05) increase in Group B rats when compared with the control rats, and a significant (p<0.05) decrease in Group C rats when compared with Group B rats.
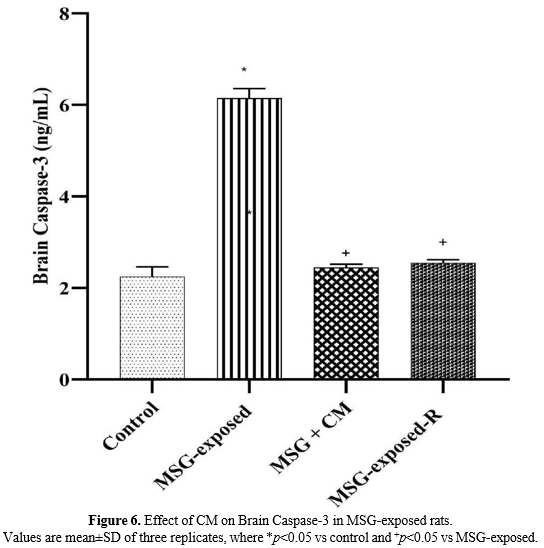
Effect of CM on Brain Caspase-3 in MSG-exposed rats
The results of brain caspase-3 are presented in Figure 6. The results revealed a significant (p<0.05) increase in Group B rats when compared with the control rats and a significant (p<0.05) decrease in Groups C and D rats when compared with Group B rats.
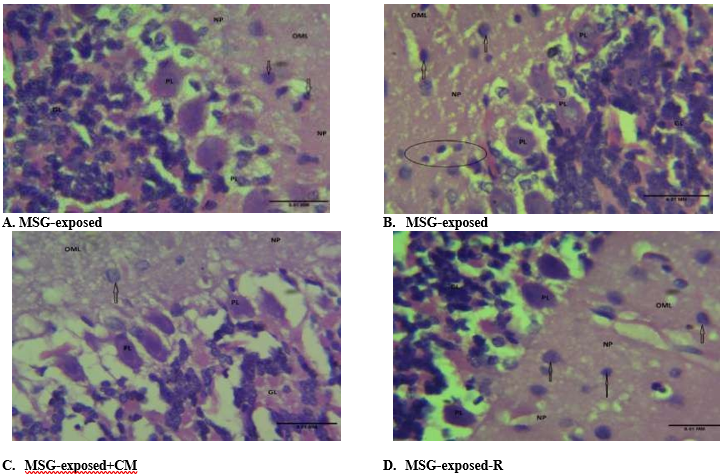
Effect of CM on Brain Histology in MSG-exposed rats
Photomicrograph (Figure 7 [A, B, C, and D]) demonstrated cerebellum histomorphology made up of its classical layers: the Grey matter consisting of the outer molecular layer (OML), Purkinje layer (PL), granular layer (GL), all dispersed within the neuropil (NP) and the White matter composed of neuronal axons and Glial cells. The blood capillaries appear normal and unremarkable. Features were consistent with normal cerebellar tissue.
Figure 7. Photomicrograph of brain sections stained by H & E (Mg×400).
Where OML=Outer molecular layer, PL=Purkinje layer, GL=Granular layer, and NP=Neuropil.
Discussion
The MSG is a commonly used flavour enhancer in food industries worldwide. However, recent neurobiological research has raised significant concerns about its potential neurotoxic effects [5,6]. The brain is particularly vulnerable to MSG-induced damage due to its high metabolic rate and limited antioxidant capacity [22]. While complete avoidance of MSG may be impractical, there is a substantial need for an effective strategy to address MSG's neurotoxic effects. Therefore, the present study investigated CM's therapeutic potential against MSG-induced pro-inflammation and oxidative neurotoxicity in rats.
The results demonstrated that MSG exposure led to substantial neurological alterations across multiple biochemical and physiological parameters. Our findings revealed that MSG-exposed rats had the least body weight change (BWC), which does not support the controversy that MSG causes obesity but is consistent with an earlier report that MSG suppresses weight gain [23,24]. Moreover, it led to a significant reduction in brain weight compared with controls, suggesting that MSG can cause metabolic disruptions and potential neuronal damage [25]. Notably, CM administration mitigated these changes and showed promising recovery in brain weight, suggesting a potential neuroprotective effect.
Oral administration of Msg (6 g/kg bw) resulted in a prominent increase in inflammatory markers, including IL-1β, TNF-α, MPO, NO, CRP, and NF-kβ in MSG-exposed rats, indicating a robust inflammatory response in the brain [26]. This inflammatory cascade is consistent with previous studies highlighting glutamate's excitotoxic potential, which can lead to neuronal damage through excessive receptor stimulation and subsequent cellular stress [27].
Oxidative stress markers further substantiated the neurotoxic effects of MSG. Its administration significantly elevated brain oxidative stress markers, particularly MDA levels, which indicate lipid peroxidation, while depleting antioxidant enzymes (CAT, SOD, GSH, GST, and GPx). These findings align with previous studies demonstrating MSG's ability to induce oxidative stress through excessive free radical generation [28,29].
Acetylcholine and dopamine play crucial roles in cognitive regulation. In the study, the neurotransmitter profile was significantly altered in MSG-exposed rats. There was a prominent increase in acetylcholinesterase (AChE) activity and a significant decrease in dopamine levels, suggesting neurotransmitter dysregulation. These changes are consistent with previous studies linking MSG consumption to cognitive impairment and neurochemical imbalances [30].
Caspase-3, an executioner protease, functions critically in the process of programmed cell death. The elevated caspase-3 activity in MSG-exposed rats further indicates an active apoptotic process, highlighting the potential for neuronal death [31]. Histological examination did not show significant alterations in cerebellar architecture with MSG exposure. This lack of structural alteration may suggest that the neurotoxic effects of MSG at the given dose and duration manifest predominantly at the molecular level before progressing to overt morphological damage, as earlier noted by Mekkawy et al. (2020), who reported that MSG administration at 6 mg/g bw led to cerebellar histological disruption after 60 days [32]. Similar observations have been reported in earlier studies, which noted the time-dose dependent nature of histological alterations, where biochemical disruptions preceded histological changes, signifying the sensitivity of biochemical assays in detecting early toxic events [33,34]. This indicates a potential window for early therapeutic intervention.
On the other hand, CM administration significantly attenuated these adverse effects. Its administration significantly abated the inflammatory response, reducing IL-1β, tumour necrosis factor-α (TNF-α), and nuclear factor-kappa B concentrations to near-control levels. This anti-inflammatory effect can be attributed to CM's rich bioactive compounds, including lactoferrins, immunoglobulins, and antioxidant proteins [35]. The milk's ability to modulate inflammatory pathways suggests a potential therapeutic mechanism in mitigating neuroinflammation.
Furthermore, treatment with CM substantially decreased the MDA levels and prominently restored the activities of antioxidant enzymes in the brain. This study indicates the antioxidant property of CM, which aligns with previous studies [36,37]. CM supplementation effectively normalised AChE activity and dopamine levels, suggesting its potential to preserve neurotransmitter function and cognitive processes. This effect may be attributed to CM's bioactive peptides and proteins, which have been shown to modulate neurotransmitter systems and support synaptic function [38]. The significant reduction in caspase-3 activity following CM administration indicates its anti-apoptotic properties, potentially through the regulation of death signalling pathways [39]. The recovery group showed partial improvement in various parameters compared with the MSG-exposed group, indicating the brain's inherent capacity for recovery. However, some markers, particularly inflammatory markers and indicators of oxidative stress, including neurotransmitter profiles, remained elevated compared to the CM-treated group. This issue suggests that natural recovery alone may be insufficient for the complete resolution of MSG-induced neurotoxicity. This observation aligns with previous studies on neural tissue recovery following toxic insult [40]. By implication, this finding suggests that CM can be a reliable and efficient treatment strategy for MSG-induced neurotoxicity.
Conclusions
In conclusion, the study suggests that CM supplementation can mitigate the neurotoxicity caused by MSG through antioxidant, anti-inflammatory, and anti-apoptotic mechanisms, potentially improving cognitive functions and integrity. Therefore, CM may serve as a therapeutic agent amenable to a diet that can neutralise MSG's neurotoxic effect.
Ethical Considerations
The Ethical Review Board of the Physiology Department approved the research protocol issued under the Ethical Approval Number EKSU/P100/2024/01/002. The globally recognised guidelines for the care and use of laboratory animals, established by the Canadian Council on Animal Care and the Guidelines for Protocol Review (NRC, 1997), were strictly followed. The experiment also complied with the National Institutes of Health guidelines for the care and use of laboratory animals.
Authors' Contributions
1. Conception/ design of the study: Dr Moshood Abiola Folawiyo and Dr Modinat Adebukola Adefisayo
2. Acquisition of the data: Dr Moshood Abiola Folawiyo, Dr Omosola Fisayo Anifowose, Mr Timilehin Micheal Oni, and Prof Ayodeji Folorunsho Ajayi
3. Analysis or interpretation of data: Dr Moshood Abiola Folawiyo, Dr Modinat Adebukola Adefisayo, Mr Timilehin Micheal Oni, and Prof Ayodeji Folorunsho Ajayi
4. Drafting of the manuscript: Dr Omosola Fisayo Anifowose and Mr Timilehin Micheal Oni
5. Critical revision of the manuscript for important Intellectual content: Dr Moshood Abiola Folawiyo, Dr Modinat Adebukola Adefisayo, and Prof Ayodeji Folorunsho Ajayi.
Acknowledgement
We would like to extend our sincere gratitude to the Laboratory Unit and Department of Physiology staff for their technical support in carrying out this work.
Conflict of Interests
The authors declare that they have no conflict of interest.
Funding
The research was fully funded by the researchers.
References
-
- Mohammad Ahmadi Soleimani S, Ekhtiari H, Cadet JL. Drug-induced neurotoxicity in addiction medicine: From prevention to harm reduction. Prog Brain Res. 2016;223:19-41. [DOI: 10.1016/bs.pbr.2015.07.004] [PMID: 26806769]
- Nabi M, Tabassum N. Role of environmental toxicants on neurodegenerative disorders. Front Toxicol. 2022;4:837579. [DOI: 10.3389/ftox.2022.837579] [PMID: 35647576]
- Spencer PS, Lein PJ. Neurotoxicity. 2014;727-39. [DOI: 10.1016/B978-0-12-386454-3.00169-X]
- Bilge S. Neurotoxicity, types, clinical manifestations, diagnosis and. neurotoxicity: new advances. 2022:173. [LINK]
- Zanfirescu A, Ungurianu A, Tsatsakis AM, Nițulescu GM, Kouretas D, Veskoukis A, et al. A review of the alleged health hazards of monosodium glutamate. Compr Rev Food Sci Food Saf. 2019;18(4):1111-34. [DOI: 10.1111/1541-4337.12448] [PMID: 31920467]
- Niaz K, Zaplatic E, Spoor J. Extensive use of monosodium glutamate: A threat to public health? EXCLI J. 2018;17:273-8. [DOI: 10.17179/excli2018-1092] [PMID: 29743864]
- Liang Q, Li D, Li J, Li Y, Zou Y, Zhang Y. Protective effect of Danshensu against neurotoxicity induced by monosodium glutamate in adult mice and their offspring. Heliyon. 2024;10(3):e25546. [DOI: 10.1016/j.heliyon.2024.e25546] [PMID: 38356496]
- Köles L, Kató E, Hanuska A, Zádori ZS, Al-Khrasani M, Zelles T, et al. Modulation of excitatory neurotransmission by neuronal/glial signalling molecules: interplay between purinergic and glutamatergic systems. Purinergic Signal. 2016;12(1):1-24. [DOI: 10.1007/s11302-015-9480-5] [PMID: 26542977]
- Turski GN, Ikonomidou C. Glutamate as a neurotoxin, handbook of neurotoxicity. 2014. [DOI: 10.1007/978-1-4614-5836-4_84]
- Ademiluyi AO, Oyeniran OH, Oboh G. Dietary monosodium glutamate altered redox status and dopamine metabolism in lobster cockroach (Nauphoeta cinerea). J Food Biochem. 2020;44(11):e13451. [DOI: 10.1111/jfbc.13451] [PMID: 32851688]
- Sreejesh PG, Sreekumaran E. Effect of monosodium glutamate on striato-hippocampal acetylcholinesterase level in the brain of male wistar albino rats and its implications on learning and memory during aging. Biosci Biotech Res Comm. 2018;11(1):76-82. [DOI: 10.21786/bbrc/11.1/11]
- Swelum AA, El-Saadony MT, Abdo M, Ombarak RA, Hussein EOS, Suliman G, et al. Nutritional, antimicrobial and medicinal properties of Camel's milk: A review. Saudi J Biol Sci. 2021;28(5):3126-36. [DOI: 10.1016/j.sjbs.2021.02.057] [PMID: 34025186]
- Wernery U. Camel milk, the white gold of the desert. J Camel Pract Res. 2006;13(1):15-26. [LINK]
- Vincenzetti S, Cammertoni N, Rapaccetti R, Santini G, Klimanova Y, Zhang JJ, et al. Nutraceutical and functional properties of camelids’ milk. Beverages. 2022;8(1):12. [DOI: 10.3390/beverages8010012]
- Ayoub MA, Palakkott AR, Ashraf A, Iratni R. The molecular basis of the anti-diabetic properties of camel milk. Diabetes Res Clin Pract. 2018;146:305-12. [DOI: 10.1016/j.diabres.2018.11.006] [PMID: 30452940]
- Singh R, Mal G, Kumar D, Patil NV, Pathak KM. Camel milk: an important natural adjuvant. Agricultur Res. 2017;6(4):327-40. [LINK]
- Thongsepee N, Martviset P, Chantree P, Sornchuer P, Sangpairoj K, Prathaphan P, et al. Daily consumption of monosodium glutamate pronounced hypertension and altered renal excretory function in normotensive and hypertensive rats. Heliyon. 2022;8(10):e10972. [DOI: 10.1016/j.heliyon.2022.e10972] [PMID: 36247159]
- Ajayi AF, Akhigbe RE. Assessment of sexual behaviour and fertility indices in male rabbits following chronic codeine use. Androl. 2020;8(2):509-15. [DOI: 10.1111/andr.12717] [PMID: 31627249]
- Sedlak J, Lindsay RH. Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman's reagent. Anal Biochem. 1968;25(1):192-205. [DOI: 10.1016/0003-2697(68)90092-4] [PMID: 4973948]
- Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR. Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Analytical Biochem. 1982;126(1):131-8. [DOI: 10.1016/0003-2697(82)90118-X]
- Shoukheba MY, Ibrahim WS. Caspase-3 activity as a novel biomarker in type 2 diabetes mellitus patients with chronic periodontitis. Egy Den J. 2016;62(3):20-31. [LINK]
- Singh M, Panda SP. The Role of monosodium glutamate (MSG) in Epilepsy and other Neurodegenerative diseases: phytochemical-based therapeutic approaches and mechanisms. Curr Pharm Biotechnol. 2023. [DOI: 10.2174/1389201024666230726161314] [PMID: 37496245]
- Rahayu MS, Wahyuni S. Effects of oral administration of monosodium glutamate (MSG) on obesity in male wistar rats (Rattus norvegicus). J Biomed Translation Res. 2021;5(9):879-82. [DOI: 10.32539/bsm.v5i9.355]
- Kondoh T, Torii K. MSG intake suppresses weight gain, fat deposition, and plasma leptin levels in male Sprague-Dawley rats. Physiol Behav. 2008;95(1-2):135-44. [DOI: 10.1016/j.physbeh.2008.05.010] [PMID: 18559279]
- Akataobi US. Effect of monosodium glutamate (MSG) on behavior, body and brain weights of exposed rats. Environ Dis. 2020;5(1):3-8. [DOI: 10.4103/ed.ed_31_19]
- Asejeje FO, Abiola MA, Adeyemo OA, Ogunro OB, Ajayi AM. Exogenous monosodium glutamate exacerbates lipopolysaccharide-induced neurobehavioral deficits, oxidative damage, neuroinflammation, and cholinergic dysfunction in rat brain. Neurosci Lett. 2024;825:137710. [DOI: 10.1016/j.neulet.2024.137710.] [PMID: 38432355]
- Lian YN, Lu Q, Chang JL, Zhang Y. The role of glutamate and its receptors in central nervous system in stress-induced hyperalgesia. Int J Neurosci. 2018;128(3):283-90. [DOI: 10.1080/00207454.2017.1387112] [PMID: 28969521]
- Al-Otaibi AM, Emam NM, Elabd HK, Esmail NI. Toxicity of monosodium glutamate intake on different tissues induced oxidative stress: A Review. J Med Life Sci. 2022;4(4):68-81. [DOI: 10.21608/jmals.2022.264345]
- Kesherwani R, Bhoumik S, Kumar R, Rizvi SI. Monosodium glutamate even at low dose may affect oxidative stress, inflammation and neurodegeneration in rats. Indian J Clin Biochem. 2024;39(1):101-9. [DOI: 10.1007/s12291-022-01077-1.] [PMID: 38223009]
- Abu-Taweel GM. Effect of monosodium glutamate and aspartame on behavioral and biochemical parameters of male albino mice. Afr J Biotechnol. 2016;15(15):601-12. [DOI: 10.5897/AJB2015.15199]
- Mansour AA, Nassan MA, Saleh OM, Soliman MM. PROTECTIVE effect of camel milk as anti-diabetic supplement: biochemical, molecular and immunohistochemical study. Afr J Tradit Complement Altern Med. 2017;14(4):108-19. [DOI: 10.21010/ajtcam.v14i4.13] [PMID: 28638873]
- Mekkawy AM, Ahmed YH, Khalaf AAA, El-Sakhawy MA. Ameliorative effect of Nigella sativa oil and vitamin C on the thyroid gland and cerebellum of adult male albino rats exposed to Monosodium glutamate (histological, immunohistochemical and biochemical studies). Tissue Cell. 2020;66:101391. [DOI: 10.1016/j.tice.2020.101391] [PMID: 32933714]
- Arrighetti F, Ambrosio E, Astiz M, Capítulo AR, Lavarías S. Differential response between histological and biochemical biomarkers in the apple snail Pomacea canaliculata (gasteropoda: amullariidae) exposed to cypermethrin. Aquat Toxicol. 2018;194:140-51. [DOI: 10.1016/j.aquatox.2017.11.014] [PMID: 29179149]
- Khalaf R, Duarte Bateman D, Reyes J, Najafali D, Rampazzo A, Bassiri Gharb B. Systematic review of pathologic markers in skin ischemia with and without reperfusion injury in microsurgical reconstruction: Biomarker alterations precede histological structure changes. Microsurgery. 2024;44(2):e31141. [DOI: 10.1002/micr.31141]
- Mirmiran P, Ejtahed HS, Angoorani P, Eslami F, Azizi F. camel milk has beneficial effects on diabetes mellitus: a systematic review. Int J Endocrinol Metab. 2017;15(2):e42150. [DOI: 10.5812/ijem.42150] [PMID: 29026408]
- Behrouz S, Saadat S, Memarzia A, Sarir H, Folkerts G, Boskabady MH. The antioxidant, anti-inflammatory and immunomodulatory effects of camel milk. Front Immunol. 2022;13:855342. [DOI: 10.3389/fimmu.2022.855342] [PMID: 35493477]
- Khan MZ, Xiao J, Ma Y, Ma J, Liu S, Khan A, et al. Research development on anti-microbial and antioxidant properties of camel milk and its role as an anti-cancer and anti-hepatitis agent. Antioxidants (Basel). 2021;10(5):788. [DOI: 10.3390/antiox10050788] [PMID: 34067516]
- Kula J. Medicinal values of camel milk. Int J Vet Sci Res. 2016;2(1):18-25. [DOI: 10.17352/ijvsr.000009]
- Singh R, Mal G, Kumar D, Patil NV, Pathak KM. Camel milk: an important natural adjuvant. Agricultur Res. 2017;6(4):327-40. [DOI: 10.1007/s40003-017-0284-4]
- Krucoff MO, Rahimpour S, Slutzky MW, Edgerton VR, Turner DA. Enhancing nervous system recovery through neurobiologics, neural interface training, and neurorehabilitation. Front Neurosci. 2016;10:584. [DOI: 10.3389/fnins.2016.00584] [PMID: 28082858]










































 , Modinat Adebukola Adefisayo2
, Modinat Adebukola Adefisayo2 

 , Omosola Fisayo Anifowose1
, Omosola Fisayo Anifowose1 

 , Timilehin Micheal Oni *3
, Timilehin Micheal Oni *3 

 , Ayodeji Folorunsho Ajayi4
, Ayodeji Folorunsho Ajayi4