Ethics code: 016/EC-04/FK-06/UNIZAR/I/2025


1- Biology Education, Department of Biology Education, Faculty of Teacher Training and Education, Universitas Muhammadiyah Sukabumi, Sukabumi, Indonesia. , ginanuranti@ummi.ac.id
2- Biology Education, Department of Biology Education, Faculty of Teacher Training and Education, Universitas Muhammadiyah Sukabumi, Sukabumi, Indonesia.
Full-Text [PDF 622 kb]
(76 Downloads)
|
Abstract (HTML) (479 Views)
Full-Text: (3 Views)
Introduction
Chloranthus erectus (C. erectus), commonly known as Karas Tulang, belongs to the Chloranthaceae family. This species is indigenous to various regions across Asia, including India, China, and several Southeast Asian countries, such as Indonesia [1].
In Indonesia, traditional communities frequently use this plant in tea form, believing it has properties that enhance stamina and help prevent fatigue [2,3]. In other countries as well, almost all parts of this plant can be utilised both as food and traditional medicine. In China, the flowers of C. erectus are used to scent tea, whereas in India, the stems serve as contraceptives and the roots are used as antispasmodics during childbirth [4].
Previous studies have indicated that C. erectus has a significant biological and pharmacological potential. C. erectus contains a wide range of phytochemicals, including alkaloids, flavonoids, and terpenoids, which contribute to its antioxidant properties, with methanol extracts exhibiting the highest activity (88.36% in leaves) [5,6]. Furthermore, leaf extracts of C. erectus have shown antibacterial effects, particularly against K. pneumoniae, as well as low antiviral activity against DENV-2 [7]. C. erectus also produces novel sesquiterpenes and diterpenes that inhibit human dihydroorotate dehydrogenase, indicating its potential in therapies related to immune modulation [8]. Other studies have also reported significant anti-inflammatory potential of C. erectus leaf extracts in rodent models [9]. Collectively, these findings suggest that C. erectus is a promising medicinal resource. However, further investigations are required.
Despite the high biological and pharmacological potential of C. erectus, its safety has not been evaluated. Acute toxicity testing of a related species, C. serratus, has yielded concerning results, indicating that plant extracts can lead to severe organ damage, particularly to the liver, kidneys, and heart, with an LD50 of 41.12 g/kg in mice, indicating a high level of toxicity [10]. These findings provide an initial indication of the potential toxicity of C. erectus.
Therefore, the present study aimed to assess the acute toxicity of C. erectus and determine its safety classification for potential use. Given the potential risks associated with C. erectus and the concerning toxicity results of its related species, it is crucial to conduct comprehensive safety evaluations before considering its use in pharmaceutical applications. These toxicity evaluations ensured a thorough understanding of the safety profile of the plant.
Materials and Methods
The primary material used was C. erectus leaves from the Waluran Village Forest in Sukabumi, West Java, Indonesia. The instruments used in this study included laboratory glassware, a Büchner funnel with a vacuum, a rotary vacuum evaporator, a macerator, a separatory funnel, a 250 mL Erlenmeyer flask, a 1 mL measuring pipette, a dissection kit, oral gavage for mice, a scalpel no.11 and no.20, 1 cc and 3 cc syringes, as well as surgical instruments. In addition, a complete set of cages with feeding and drinking equipment was employed.
The acute toxicity test was conducted using female Swiss Webster mice (Mus musculus) aged 6-8 weeks, each weighing 20-30 grams and not pregnant, with a total of 16 mice used [11]. Before use, the mice underwent an acclimatization process for five days prior to the administration of the test substance. During this period, the mice were placed in standard laboratory cages under controlled environmental conditions, including temperature, humidity, and a 12-hour light-dark cycle. The mice were given standard feed and drinking water ad libitum to ensure physiological stability and to minimize the effects of external stress factors during the testing phase. The mice underwent a fasting period of 14–18 h, during which they were allowed to access water. After the fasting phase, each mouse was weighed and administered a single dose of the test substance via gavage needle. Feeding was reintroduced approximately 3–4 h after administration of the test substance [12].
Preparation of Methanol Extract of Chloranthus erectus Leaves
Chloranthus erectus was collected from the Waluran Village Forest, Waluran District, Sukabumi Regency (Figure 1). The plant was cleaned and dried in a shaded area until thoroughly dried. Once dry, the sample was ground using a blender to obtain leaf powder.
 Figure 1. Morphological features of Chloranthus erectus collected from Waluran Village Forest, Sukabumi, West Java, Indonesia
Figure 1. Morphological features of Chloranthus erectus collected from Waluran Village Forest, Sukabumi, West Java, Indonesia
Extraction was carried out using the maceration method, where 200 g of leaf powder was soaked in methanol for 3 × 24 h. The resulting macerate was filtered using a Büchner funnel and concentrated using a rotary vacuum evaporator at a temperature of approximately 50°C, resulting in 11.56 g of C. erectus extract.
Toxicity Method Test
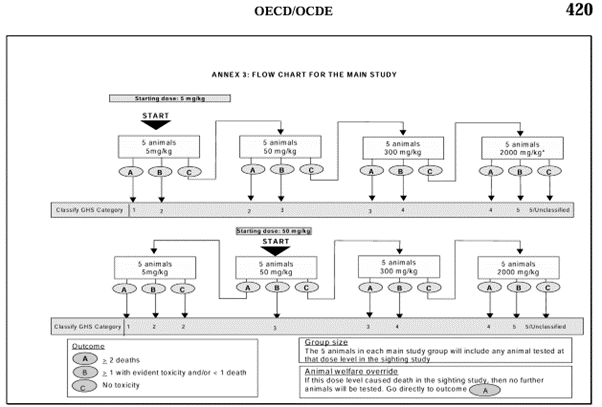
The acute toxicity test was performed according to the Organization for Economic Co-operation and Development (OECD) Test Guideline 420: Acute Oral Toxicity-Fixed Dose procedure guidelines (Figure 2). A preliminary test was performed to determine the initial dose, using one mouse per dose. The dose was decreased if mortality occurred, and increased if no signs of toxicity were observed. The preliminary test was stopped if signs of toxicity, such as altered autonomic activity (including crawling), diarrhea, nasal discharge, self-injurious behavior, and bleeding from body openings, were observed, or if no toxic effects were observed at a dose of 2000 mg/kg body weight. In the preliminary test, a dose of 50 mg/kg body weight was administered based on previous acute toxicity testing on a species of the same genus, C. serratus, which indicated hazardous results with an LD₅₀ of 41.12 g/kg in mice [10].
In the main test, five mice were used for each dose level: 5, 50, 300, and 2000 mg/kg body weight. The initial dose, determined during the preliminary test, was administered after measuring the body weight of the mice. Body weight was recorded prior to treatment and at the time of dissection at the end of the observation period to monitor changes in body weight during the testing period. Post-administration, intensive observation was conducted at 30-minute intervals for the first 4 h, every 4 h for the subsequent 24 h, and once daily for 14 days. If no mortality occurred, the dose would be increased to the next level. In the absence of mortality, the dosage was increased to the next level. Conversely, if ≥2 mice succumbed, the dosage was reduced to the preceding level. If ≥1 sign of toxicity and/or ≤1 death was observed, the LD₅₀ range could be directly determined [13]. The primary indicator of mortality was rigor mortis, with additional indicators including the absence of a pulse, lack of respiratory movements, no response to stimuli (such as pinching), and grayish mucous membranes. The main test was conducted in parallel with the control group, each consisting of five mice observed for 14 days. On day 14, all mice were euthanized via cervical dislocation, and visceral organs were isolated for organ weight index calculation.
Observation of Toxicity Symptoms
Comprehensive observations were conducted over 24 hours at intervals of 0, 0.5, 4, 8, 12, 16, 20, and 24 h, and subsequently once daily until the 14th day following the administration of C. erectus extract to identify toxic symptoms. The recorded toxic symptoms included behavioral changes and physical responses, such as motor activity, Straub tail reaction, piloerection, ptosis, and pupil and corneal reflexes. Additionally, observations included lacrimation, catalepsy, body posture, hanging behavior, recovery, flexion, and Haffner reflex. Other recorded symptoms included mortality, grooming, defecation, urination, breathing, salivation, vocalization, tremors, seizures, and twisting movements [12]. Each observation interval lasted five minutes to monitor the appearance of toxic symptoms in the test animals.
Mice Weight Data Analysis
After the administration of C. erectus extract, the mice were monitored for 24 h. The body weights of the mice were recorded from days 1 to 14, provided that no deaths occurred. Changes in body weight before and after the administration of C. erectus extract were analyzed within each group and subsequently compared between the control and experimental groups.
Relative Organ Weight Data Analysis
The relative weights of the visceral organs of the mice, including the heart, kidney, liver, spleen, stomach, and ovary, were measured using the following formula:
Relative organ weight %=Organ weight gramsBody weight of animal gramsx 100%
Results
Effect of C. erectus Extract on the Mortality of Mice
In the preliminary phase, a dose of 50 mg/kg was administered to the test animals. All test animals were observed for 24 h after administration of the dose. The results revealed that there were no deaths among the mice, and no significant signs of toxicity were observed. This finding indicates that the 50 mg/kg dose was relatively safe and did not cause acute toxic effects in the test animals. According to the principles of toxicity testing, if the initial test dose (50 mg/kg) results in no adverse effects, further testing can be conducted using higher doses to assess the toxicity threshold.
After the 300 mg/kg dose, which also resulted in no mortality, the study was advanced to a higher dose of 2000 mg/kg. This dose is considered sufficiently high and is commonly used to assess the toxicity threshold of a compound by using a fixed-dose procedure [14]. The results also showed no deaths among the test animals.
Effect of C. erectus Extract on Behavioral Changes in Mice
Behavioral and physiological responses in mice were observed on days 1, 7, and 14 following oral administration of C. erectus extract at doses of 300 mg/kg and 2000 mg/kg body weight (Table 1). Throughout all observation periods, no abnormal behavioral changes or toxic symptoms were recorded for any of the assessed parameters, especially the most toxic symptoms. The parameters included pupillary and corneal reflexes, grooming, posture, respiration, vocalization, and urination, all of which were within normal limits. No signs of autonomic or central nervous system toxicity were observed in either group. However, there was a notable increase in motor activity in the 2000 mg/kg group, whereas the 300 mg/kg group maintained normal motor activity levels throughout the study. Overall, the data consistently indicated the absence of observable toxicity-related behavior across both tested dose levels and time points.
Effect of C. erectus Extract on Body Weight in Mice
Body weight measurements were conducted both before and after treatment to assess the effect of C. erectus extract on weight gain in mice. As demonstrated in Table 2, there were numerical differences in weight gain among the groups compared to the control group. Generally, the groups receiving 300 mg/kg and 2000 mg/kg showed lower body weight gain relative to the control group. However, statistical analysis using the Kruskal-Wallis test revealed no significant intergroup differences (P=0.651). This finding suggests that the extract did not induce statistically significant changes in body weight within the tested dose range.
Effect of C. erectus Extract on Relative Organ Weight in Mice
Observations of organ weights were conducted after a 14-day observation period, during which toxicity symptoms in the test animals were monitored. After this period, the animals were dissected to collect several vital organs, including the heart, kidneys, liver, spleen, stomach, and ovary. The weight of each organ was recorded to assess its relative weight. Figure 2 presents the mean±SD of visceral organ weight indices in mice treated with C. erectus extract at various doses.
Table 1. Effect of C. erectus extract on behavioral changes in mice
| Toxic Symptoms |
Dose 300 mg/kg BW |
Dose 2000 mg/kg BW |
| Day 1 |
Day 7 |
Day 14 |
Day 1 |
Day 7 |
Day 14 |
| Mortality |
None |
None |
None |
None |
None |
None |
| Motor activity |
Normal |
Normal |
Normal |
Increase |
Increase |
Increase |
| Straub tail reaction |
None |
None |
None |
None |
None |
None |
| Piloerection |
None |
None |
None |
None |
None |
None |
| Ptosis |
None |
None |
None |
None |
None |
None |
| Pupillary reflex |
Normal |
Normal |
Normal |
Normal |
Normal |
Normal |
| Corneal reflex |
Normal |
Normal |
Normal |
Normal |
Normal |
Normal |
| Lacrimation |
None |
None |
None |
None |
None |
None |
| Catalepsy |
None |
None |
None |
None |
None |
None |
| Posture |
Normal |
Normal |
Normal |
Normal |
Normal |
Normal |
| Hanging behavior |
Normal |
Normal |
Normal |
Normal |
Normal |
Normal |
| Re-establishment |
Normal |
Normal |
Normal |
Normal |
Normal |
Normal |
| Flexion |
Normal |
Normal |
Normal |
Normal |
Normal |
Normal |
| Haffner's reflex |
Normal |
Normal |
Normal |
Normal |
Normal |
Normal |
| Grooming |
Normal |
Normal |
Normal |
Normal |
Normal |
Normal |
| Defecation |
Normal |
Normal |
Normal |
Normal |
Normal |
Normal |
| Urination |
Normal |
Normal |
Normal |
Normal |
Normal |
Normal |
| Respiration |
Normal |
Normal |
Normal |
Normal |
Normal |
Normal |
| Salivation |
None |
None |
None |
None |
None |
None |
| Vocalization |
Normal |
Normal |
Normal |
Normal |
Normal |
Normal |
| Tremor |
None |
None |
None |
None |
None |
None |
| Convulsions |
None |
None |
None |
None |
None |
None |
| Writhing |
None |
None |
None |
None |
None |
None |
Table 2. Effect of C. erectus extract on body weight in mice
| Group |
Initial Body Weight (g) |
Final Body Weight (g) |
Body Weight Gain (g) |
P-value |
| Control |
17.60±1.342 |
25.30±2.289 |
7.7±1.905 |
0.651 |
| 300 mg/kg BW |
22.78±1.401 |
25.35±1.026 |
2.57±1.836 |
| 2000 mg/kg BW |
21.76±1.687 |
26.69±2.021 |
5.196±0.786 |
Figure 2. Flow chart for the main study of OECF 420
Over 14 days, oral administration of Chloranthus erectus methanol extract at dosages of 300 mg/kg BW and 2000 mg/kg BW did not produce consistent trends in the visceral organ weight index across the evaluated organs. In the liver and kidneys, mice receiving 300 mg/kg and 2000 mg/kg showed slightly elevated organ indices compared to the control group. Specifically, liver index values increased from 0.046 g/g BW in the control group to 0.065 g/g BW in the 300 mg/kg group, then decreased to 0.061 g/g BW in the 2000 mg/kg group. In contrast, the spleen and heart indices showed a slight reduction in the treated groups compared with the control, whereas the ovarian indices remained virtually unchanged across all groups (0.00004 g/g BW). The observed variation in standard deviations suggests biological variability rather than a dose-dependent effect. Statistical analysis using one-way ANOVA revealed no significant differences in organ weight indices among the groups (P>0.05 for all organs), confirming the absence of treatment-related pathological hypertrophy or atrophy in the examined organs.
Discussion
The present research aimed to evaluate the acute oral toxicity profile of Chloranthus erectus methanol extract in mice using the OECD 420 fixed-dose procedure. Oral administration of the extract at doses of 300 mg/kg and 2000 mg/kg body weight caused no mortality or observable signs of systemic toxicity throughout the 14-day observation period. Moreover, body weight gain was not significantly affected, and the relative weights of major visceral organs, including the liver, kidneys, heart, spleen, stomach, and ovaries, did not differ significantly between the treated and control groups (P>0.05). These findings suggest that the methanol extract of C. erectus is relatively safe and does not produce overtly toxic effects under the tested conditions.
Various species within this genus, such as C. anhuiensis and C. japonicus, have traditionally been used in medicine to treat wounds, swelling, rheumatic pain, and infections, demonstrating a relatively good safety profile for use [15,16]. However, there is variability in toxicity levels among species within this genus, as observed in Chloranthus serratus. C. serratus has a relatively high LD₅₀ value of 41.12 g/kg in mice, indicating its significant toxicity [10]. In animal studies, extracts from the stems and leaves of C. serratus have been found to cause hepatotoxicity, as demonstrated by increased liver enzyme levels, oxidative stress, ultrastructural changes, lipid accumulation, and histological damage [17]. These toxic effects are attributed to the presence of abundant terpenoids, such as acolamone, chloranthalactone, shizukanolide, zederone, and triterpenoids, compounds often linked to hepatotoxic mechanisms [17].
In contrast, the C. erectus extract did not induce mortality or systemic toxicity at doses up to 2000 mg/kg BW. The Chloranthus genus generally contains a diverse array of phytochemicals, such as terpenoids, coumarins, lignans, simple phenylpropanoids, flavonoids, organic acids, amides, and eight other compounds [1]. These compounds predominantly exhibit biological activities, such as antioxidant, anti-inflammatory, antibacterial, and anticancer properties, while maintaining a low toxicity profile across most Chloranthus species. Specifically, phytochemical analyses of C. erectus have indicated that it primarily contains flavonoids, tannins, and saponins, which are not strongly associated with hepatotoxicity [18]. This composition may contribute to its observed protective effects, particularly its antioxidant and anti-inflammatory activities, while minimizing the risk of acute toxicity [5,6]. These findings are consistent with the traditional use of C. erectus as an herbal remedy for various health complaints when administered orally, reinforcing the assumption that its extract is relatively safe for acute use at the tested dose [16].
Behavioral assessments indicated a dose-dependent increase in motor activity in mice administered the highest dose of C. erectus extract. Although no additional neurobehavioral abnormalities, such as convulsions, tremors, or autonomic disturbances, were observed, this hyperactivity may not necessarily signify severe toxicity. Instead, it may suggest a potential stimulant effect of certain bioactive compounds present in the extract. Several phytochemicals identified in C. erectus, including dextroamphetamine, idebenone metabolite (QS-10), and pimozide, are known to modulate central nervous system activity [19]. Notably, dextroamphetamine is a well-characterized psychostimulant that increases synaptic dopamine and norepinephrine by promoting their release and inhibiting reuptake, commonly resulting in increased locomotor activity and behavioral stimulation [20,21]. Therefore, the observed increase in motor activity may reflect CNS hyperexcitability, a hallmark feature of neurostimulant exposure in acute toxicity paradigms, rather than a manifestation of irreversible neurotoxic damage.
The absence of mortality and significant physiological or behavioral disturbances at the limit dose suggests that C. erectus extract is not acutely toxic and demonstrates a substantial safety margin for oral exposure. According to the Globally Harmonized System (GHS) for the classification of chemicals, substances that show no mortality at 2000 mg/kgBW are classified under Category 5 or remain unclassified for acute toxicity [22]. Therefore, the current findings confirm the relative safety of C. erectus under the conditions of this study and provide a scientific basis for its continued investigation as a traditional medicine.
Conclusions
The methanol extract of C. erectus showed no evidence of acute oral toxicity in female mice at doses of up to 2000 mg/kg body weight. This conclusion is supported by the absence of mortality, behavioral abnormalities, and significant changes in body or relative organ weight. These results suggest that the extract has a favorable safety profile and may be classified as having low acute toxicity according to the OECD and GHS guidelines. Further investigations, including subchronic toxicity assessments, histopathological evaluations, and biochemical analyses, are necessary to confirm the long-term safety and potential therapeutic applications of C. erectus.
Ethical Considerations
This study received ethical approval from the medical and health research ethics committee of the Faculty of Medicine, Al-Azhar University Mataram, with the approval number 016/EC-04/FK-06/UNIZAR/1/2025.
Authors' Contributions
Gina Nuranti contributed to the conceptualization of the research, supervision, guidance of the toxicity testing, data analysis, and interpretation. Jujun Ratnasari was responsible for preparing extracts, analyzing, and interpreting data. Irgi Indrawan and Risti Mulyani Nur ‘Kauthsar were involved in the experimental procedures and data analysis. All authors contributed to the writing and revision of the manuscript and approved the final version for submission.
Conflict of Interests
The authors have no conflict of interest related to this study.
Funding
This research was funded by RisetMu Program on Penelitian Fundamental Reguler I Scheme, DIKTILITBANG Muhammadiyah, Indonesia.
Acknowledgement
The authors acknowledge the Biology Education Study Program and the Institute for Research and Community Service (LPPM) at Universitas Muhammadiyah Sukabumi, Indonesia, for their support. This research was funded by the RisetMu Program under the Fundamental Research Scheme (Regular I), Directorate of Higher Education, Research, and Development, Muhammadiyah.
References
- Wang AR, An H, Luo X, Song H, Dong J, Huang Q. Secondary metabolites of plants from the genus chloranthus: chemistry and biological activities. Chem Biodivers. 2015;12(4):451-73. [DOI: 10.1002/cbdv.201300376] [PMID: 25879494]
- Herlina N, Adhya I, Hendrayana Y, Nurdin N, Supartono T, Ibrahiim M, et al. Pemberdayaan masyarakat melalui penyuluhan sebagai upaya peningkatan pemahaman tumbuhan obat pada masyarakat desa cimara. BERNAS: J Pengabdian Kepada Masyarakat. 2025;6(1):696-700. [DOI: 10.31949/jb.v6i1.10488]
- Iskandar J. Pandemics and sustainable human nature relations: a case study in Baduy community, South Banten, Indonesia. E3S Web of Conf. 2021;249:01001. [DOI: 10.1051/e3sconf/202124901001]
- Tropical Plants Database. Cornwall. 2024. [Link]
- Zemry IH, Hasan NA, Hasbullah NI, Nawahwi MZ, Azzeme AM, Siti Noor DA, et al. Antioxidant potential of Chloranthus erectus (Chloranthaceae) from various solvents extract. J Exp Biol Agric Sci. 2023;11(1):75-80. [DOI: 10.18006/2023.11(1).75.80]
- Hasan NA, Ariffin S, Mohamad Azzeme A, Hasbunallah NI, Zaini M, Nawahwi A, et al. Preliminary phytochemical screening of medicinal herb, sambau paya (Chloranthus erectus). Mater Today Proc. 2023;88(2):6-9. [DOI: 10.1016/j.matpr.2023.01.365]
- Hasan NA, Wazir NN, Zainal-Abidin N, Nawahwi MZ, Badrol NA, Yasin Y, et al. Antibacterial and antiviral potential of zirconium oxide nanoparticles using extract of chloranthus erectus leaf. Asian J Chem. 2024;36(7):1705-12. [DOI: 10.14233/ajchem.2024.31775]
- Yang Q, Jia A, Liu X, Han S, Fan S. Chemical constituents from chloranthus elatior and their inhibitory effect on human dihydroorotate dehydrogenase. Planta Med. 2022;88(6):455-65. [DOI: 10.1055/a-1449-2642] [PMID: 33902131]
- Tag H, Namsa ND, Das AK, Kalita P, Mandal SC. Evaluation of anti-inflammatory potential of Chloranthus erectus (Buch.-Ham.) Verd. leaf extract in rats. J Ethnopharmacol. 2009;126(2):371-4. [DOI: 10.1016/j.jep.2009.08.015] [PMID: 19703538]
- Zhang W, Zhu JH, Cheng LB, Li YH. Experimental pathological study of acute intoxication by Chloranthus serratus Roem. Et Schalt. Fa Yi Xue Za Zhi. 2006;22(1):15-7. [PMID: 16524177]
- Badan pengawas obat dan makanan republik indonesia. peraturan kepala badan pengawas obat dan makanan Nomor 7 Tahun 2014 tentang pedoman uji toksisitas nonklinik secara in vivo. Jakarta: BPOM. 2014. [LINK]
- Badan pengawas obat dan makanan republik indonesia. panduan penyusunan protokol uji praklinik: uji toksisitas akut. jakarta: direktorat registrasi obat tradisional, suplemen kesehatan, dan kosmetik. Bpom Ri. 2022. [LINK]
- No OT. 420: Acute oral toxicity-fixed dose procedure. OECD guidelines for the testing of chemicals, Section. 2002;4:1-4. [LINK]
- Abrori C, Nurfadhila K, Nurus Sakinah E. Uji toksisitas akut ekstrak etanol daun kemangi (Ocimum sanctum) diukur dari nilai LD50 dan histopatologi ginjal acute. J Agromed Med Sci. 2019;5(1). [LINK]
- Xu J, Zhu HL, Zhang J, Du T, Guo EY, Liu WY, et al. Sesquiterpenoids from chloranthus anhuiensis with neuroprotective effects in PC12 cells. J Nat Prod. 2018;81(6):1391-8. [DOI: 10.1021/acs.jnatprod.7b01076] [PMID: 29775304]
- Liu YY, Li YZ, Huang SQ, Zhang HW, Deng C, Song XM, et al. Genus chloranthus: a comprehensive review of its phytochemistry, pharmacology, and uses. Arab J Chem. 2022;15(8):104260. [DOI: 10.1016/j.arabjc.2022.104260]
- Sun S, Wang Y, Du Y, Sun Q, He L, Zhu E, et al. Oxidative stress-mediated hepatotoxicity in rats induced by ethanol extracts of different parts of Chloranthus serratus. Pharmacogn Mag. 2020;58(1):1277-89. [DOI: 10.1080/13880209.2020.1859552] [PMID: 33355514]
- Kujur, R. S., & Singh, V. Antioxidant potential of chloranthus erectus (chloranthaceae) from various solvent extracts. J Exp Biol Agric Sci. 2023;11(1):75–80. [DOI: 10.18006/2023.11(1).75.80]
- Jeeno P, Yadoung S, Yana P, Hongsibsong S. Phytochemical profiling and antioxidant capacity of traditional plants, northern Thailand. Plants. 2023;12(23):3956. [DOI: 10.3390/plants12233956]
- Heal DJ, Smith SL, Gosden J, Nutt DJ. Amphetamine, past and present – a pharmacological and clinical perspective. J Psychopharmacol. 2013;27(6):479-96. [DOI: 10.1177/0269881113482532]
- Clemow DB, Walker DJ. The potential for misuse and abuse of medications in ADHD: A review. Postgrad Med. 2014;126(5):64-81. [DOI: 10.3810/pgm.2014.09.2801] [PMID: 25295651]
- United Nations. Globally harmonized system of classification and labelling of chemicals (GHS). Revision 8. Perserikatan Bangsa-Bangsa. 2019. [LINK]
Type of Study:
Research |
Subject:
General