Introduction
Natural products are a valuable source of medicinal agents [1]. Graviola, extensively used in traditional medicine, exhibits therapeutic properties, including anti-inflammatory, antimicrobial, antioxidant, and cytotoxic effects against tumor cells [2]. However, its potential toxicity raises significant concerns. Studies have demonstrated that Graviola extracts, while effective against cancer cells, may also harm healthy cells [1].
Toxicological research highlights acute effects, including impaired survival and cardiac function in zebrafish embryos exposed to Graviola extracts, emphasizing the need for precise dosage control (Daddiouaissa et al., 2020) [3]. Additionally, chronic consumption of Graviola has been linked to neurotoxicity, notably an atypical form of Parkinson’s disease unresponsive to standard treatments, attributed to acetogenins, a class of neurotoxic compounds [4-6]. This duality of therapeutic potential and toxicity underscores the importance of evaluating Graviola's safety profile before its broader application.
β-glucans and arabinogalactans are natural polysaccharides that have been extensively studied for their immunomodulatory properties and beneficial effects on health. However, despite their well-documented advantages, their potential toxicity remains an important area of interest for the scientific community. β-glucans, primarily found in the cell walls of cereals, mushrooms, and yeast, are known to stimulate immune responses [7]. Nevertheless, some studies suggest that they could also cause adverse effects under specific conditions, particularly by triggering excessive activation of the immune system [8]. Similarly, arabinogalactan, a polysaccharide extracted from plants, such as larch, is recognized for its prebiotic properties and its ability to stimulate immunity [9]. However, its long-term safety and toxic potential require further evaluation, especially regarding its interactions with immune receptors and its impact on intestinal balance.
Materials and Methods
We selected Graviola and a mixture of immunostimulants based on preliminary screening results, where only these two natural substances showed notable anticancer activity. To support the development of a safe dietary supplement, we conducted a subchronic toxicity study focusing particularly on reproductive effects. This was crucial because Graviola, unlike the immunostimulants, induced significant reproductive impairment in mice. The purpose was not to compare the two substances pharmacologically but to confirm the specific reproductive toxicity associated with Graviola.
In accordance with standard guidelines for subchronic toxicity testing in rodents, which typically use a 90-day (3-month) exposure period (OECD TG 408; FDA Redbook 2000; ICCF guidance), our subchronic study in mice was conducted over approximately three months.
Animals and Housing Conditions
In this study, 36 Swiss albino mice (18 males and 18 females) aged three weeks were used. These animals were supplied by the Pasteur Institute of Algiers, Algeria. Upon arrival, the mice had a body weight ranging between 17 g and 20 g. They were housed in a controlled animal facility, where environmental conditions (temperature, humidity, light-dark cycle) and nutritional standards were maintained. A two-week acclimatization period was observed prior to the start of the experiment. The mice were fed a standard pellet diet provided by ONAB (Annaba, Algeria) and given water ad libitum. They were maintained under standard housing conditions at a temperature of 25±4°C. The body weights of the animals were recorded weekly throughout the study.
The female mice were dedicated to the reproductive toxicity experiment.
Tested Products and Preparation
Two substances that were evaluated in this study are as follows:
Graviola Leaf Powder (Annona muricata): The fine powder of Graviola leaves, originating from Peru and produced in Germany, was employed. The product, commercially available in capsule form (lot C17030059, Zein Pharma), is recommended for human consumption at a dosage of two to three 500-mg capsules per day. For animal experimentation, the powder was extracted from the capsules and prepared in a form suitable for oral administration.
β-Glucan and Arabinogalactan Mixture: β-glucan, extracted from Saccharomyces cerevisiae yeast, and arabinogalactan, derived from larch tree, were obtained from Now Foods (lot no. 21046780740, Illinois, USA). This mixture, known for its immunostimulant properties, was provided in capsules, with each capsule containing 250 mg of β-glucan and 200 mg of arabinogalactan. The powder was extracted from the capsules and prepared in a form suitable for oral administration.
Experimental Design
The overall experimental design can be summarized in the following diagram:
Male mice: The mice were then randomly divided into three groups (n=6 per group).
| [Cage Control (C)] |
[Cage Graviola (G)] |
[Cage Immunostimulants (IS)] |
| ♂♂♂♂♂♂ |
♂♂♂♂♂♂ |
♂♂♂♂♂♂ |
| used for the subchronic toxicity experiment |
Female mice: The mice were then randomly divided into three groups (n=6 per group).
| [Cage Control (C)] |
[Cage Graviola (G)] |
[Cage Immunostimulants (IS)] |
| ♀♀♀♀♀♀ |
♀♀♀♀♀♀ |
♀♀♀♀♀♀ |
| used for the reproductive toxicity experiment |
Mating occurring four weeks after the start of the treatment:
C × C G × G IS × IS
Subchronic toxicity experiment:
The male mouse groups were divided as follows:
Group Control (C): The mice received only water via gastric gavage throughout the experiment.
Group Graviola (G): The mice were administered, via gastric gavage, a dose 10 times the normal dose used in the traditional pharmacopoeia for Graviola leaf powder dissolved in water. The reference dose being X=0.53 mg/mouse, they received 10*X=5.3 mg/mouse, assuming an average weight of 25 g per mouse (i.e., 212 mg/kg).
Group Immunostimulants (IS): The mice received, via gastric gavage, a dose 10 times the standard dose used in the traditional pharmacopoeia of the immunostimulant mixture (Saccharomyces cerevisiae β-glucan and arabinogalactan) dissolved in water. With a reference dose of X=0.64 mg/mouse, they received 10*X=6.4 mg/mouse, assuming an average weight of 25 g per mouse (i.e., 265 mg/kg).
After 13 weeks of experimentation on the subchronic toxicity of Graviola powder as well as the β-glucan and arabinogalactan mixture [10], the mice were euthanized by decapitation, followed by blood collection, and then the immediate shipment of the samples for biochemical testing.
All sacrificed male mice were dissected to collect the liver and brain, while for female mice (Control and G groups), the uterus was also removed. The organs were divided into two parts: one was preserved in a 10% formalin solution for future histological examination, while the other was used for oxidative stress analysis.
Measurement of Plasma Biochemical Biomarkers
The assessment of biochemical parameters related to subchronic toxicity was performed using an automated system (MINDRAY). The analyzed biomarkers include glucose, triglycerides, cholesterol, aspartate aminotransferase (ASAT), and alanine aminotransferase (ALAT). ("Biochemical analyses were performed using kits designed for human clinical use. Physiological differences between humans and mice may affect measurement accuracy, requiring cautious interpretation of the results.")
Histological Analysis
Organ samples were promptly fixed in 10% formalin solution. They were then dehydrated through a series of ethanol baths, followed by a clearing step with toluene, and embedded in paraffin at 56–58°C. Thin sections of 5 μm were obtained using a microtome (Leica RM2235). Staining was performed following the Hematoxylin-Eosin (H&E) technique. Finally, the stained sections were examined and photographed using an optical microscope (Leica, Germany).
Preparation of Organ Homogenates for Oxidative Stress
After removal and washing in 0.9% NaCl solution, the organs (0.8 g of liver or 0.25 g of brain) were processed at 4°C by grinding in 4 ml of phosphate-buffered saline (TBS: 50 mM Tris, 150 mM NaCl, pH 7.4). Once homogenized, the resulting cell suspensions were centrifuged at 9000 rpm for 15 minutes at 4°C. The supernatants were then aliquoted and stored at –20°C for later analysis.
The measured oxidative stress parameters include catalase (CAT), glutathione peroxidase (GPx), reduced glutathione (GSH), and malondialdehyde (MDA).
Reproductive Toxicity
The female control groups, as well as those treated with Graviola and immunostimulants, underwent the same subchronic toxicity protocol described above, in parallel with the male groups.
One month after the start of treatment for both sexes, mating was initiated between male and female mice of each similar group (C × C G × G IS × IS). The males were separated from the females one week after the beginning of mating, while gastric administration of Graviola or immunostimulants continued for all treated groups until the females gave birth. The newborn mice were then counted for each female group.
Data were analyzed using Microsoft Excel software for statistical calculations.
Results
Body Weight Variation
The results indicate no significant difference in body weight changes between mice treated with crude Graviola extract or immunostimulants and the control group (Figure 1).
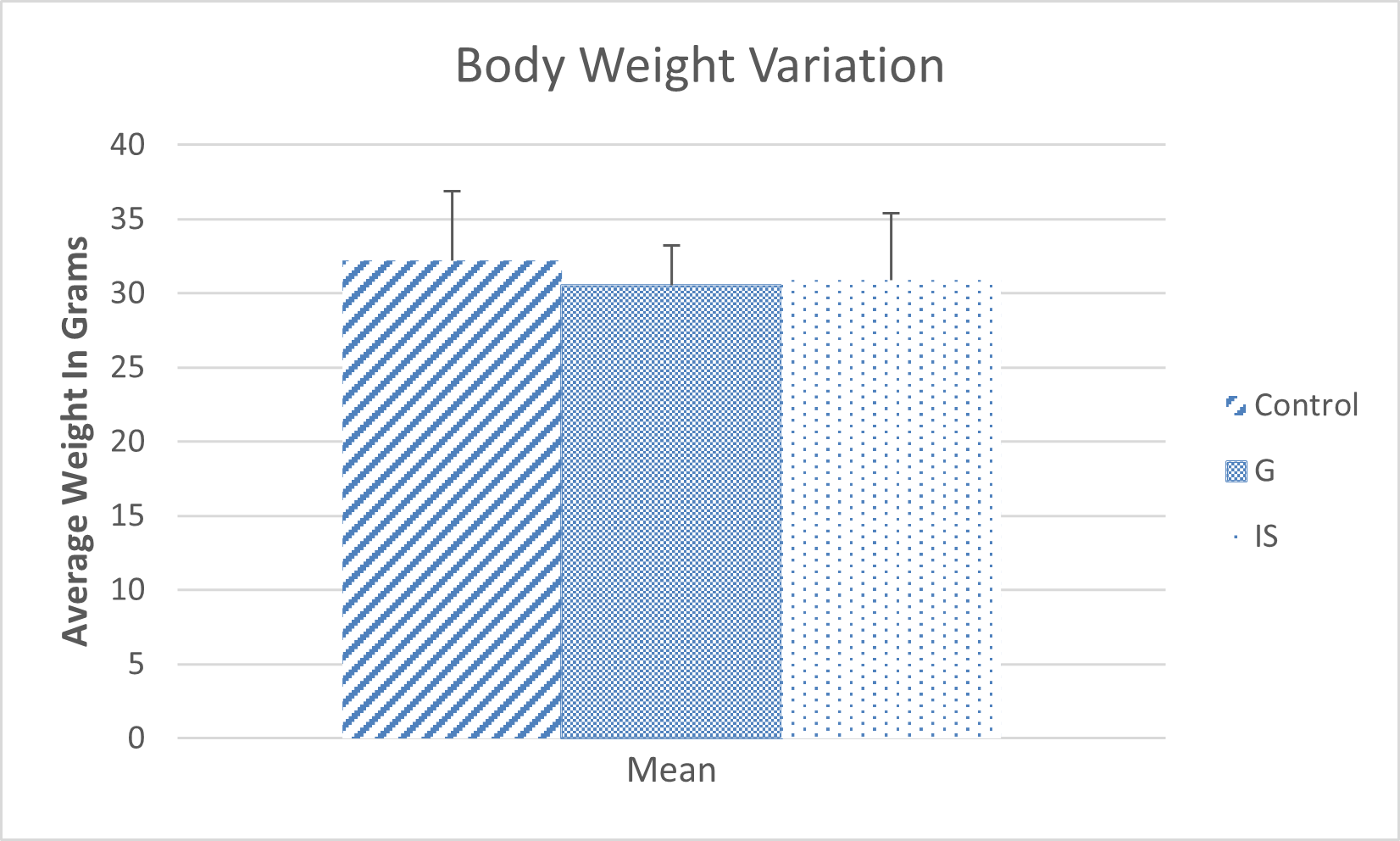
Figure 1. Variations in the average weight of mice.
Complete Blood Count (CBC)
We observed a highly significant increase in the percentage of lymphocytes (+25.24%) (Figure 2). Graviola appears to have a notable effect on red blood cell and monocyte levels (Figure 3). Specifically, we recorded a significant increase in monocytes (+40.47%) and red blood cells (+53.38%) compared to the control group. In the group treated with immunostimulants, significant increases in white blood cell levels (+24.88%) and lymphocyte levels (+29.66%) were observed compared to the control group.
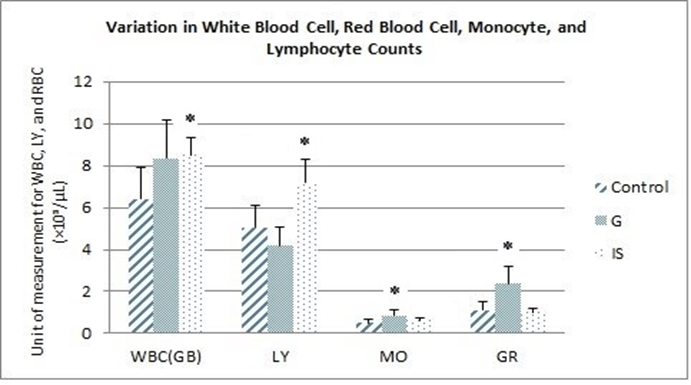
Figure 2. Variation in the blood cell count formula for white blood cells (WBC), red blood cells (GR), monocytes (MO), and lymphocytes (LY) (*p≤0.05 vs. control).
Biochemical Parameters
Blood Glucose, Cholesterol, and Triglycerides
The data presented in Figure 4 indicate a significant decrease in plasma cholesterol levels (-45.06%) in mice treated with immunostimulants compared to the control group. Additionally, the plasma concentration of blood triglycerides showed a highly significant reduction (-49.04%) in mice that received the Graviola-based treatment. However, no significant difference in blood glucose levels was observed among the three groups.
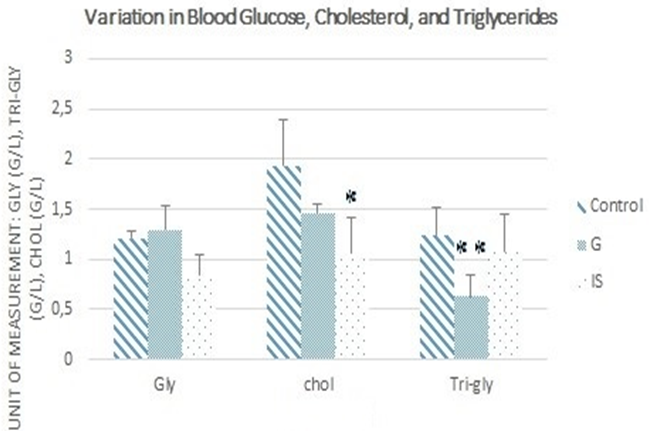
Figure 4. Variations in blood glucose (Gly), cholesterol (chol), and triglyceride (Tri-gly) levels (*p≤0.05 vs. control, **p≤0.01 vs. control).
Transaminases
For both Graviola and immunostimulants, the results show no significant difference in Aspartate Aminotransferase (AST) levels between treated mice and the control group. However, Alanine Aminotransferase (ALT) levels decreased significantly in mice treated with immunostimulants compared with the control group (Figure 5).
Histological Analyses
Graviola
Histological analysis of the liver, brain, and uterus (Figure 6) in mice treated with Graviola revealed no structural alterations compared to the control group. Hepatic and brain cells remained intact, and the overall tissue architecture appeared normal. In the uterus, no significant differences were observed between the groups, except for natural variations in endometrial thickness due to the mice's estrous cycles. No lesions or inflammatory immune cell infiltration were detected in the different uterine layers.
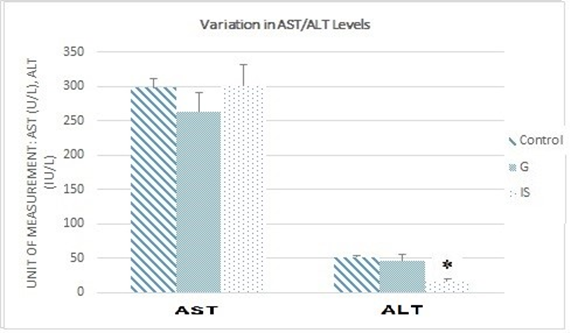
Figure 5. Variations in AST and ALT levels (*p≤0.05 vs. control).
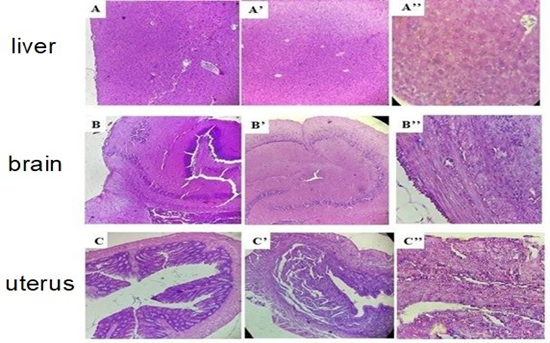
Figure 6. Histological sections of the liver, brain, and uterus in the group treated with Graviola leaf powder: A, B, C) control (×10); A’, B’, C’) treated (×10); A’’, B’’, C’’) treated (×40). Optical microscope Leica (×10) and (×40).
Immunostimulants
Given that the reproductive toxicity results in mice treated with immunostimulants showed no significant differences in litter and offspring numbers compared to the control group, the present study focused on the potential toxicity of immunostimulants on the liver and brain.
Histological analysis (Figure 7) revealed no alterations or lesions in the hepatic and brain tissues of mice treated with immunostimulants compared to the control group.
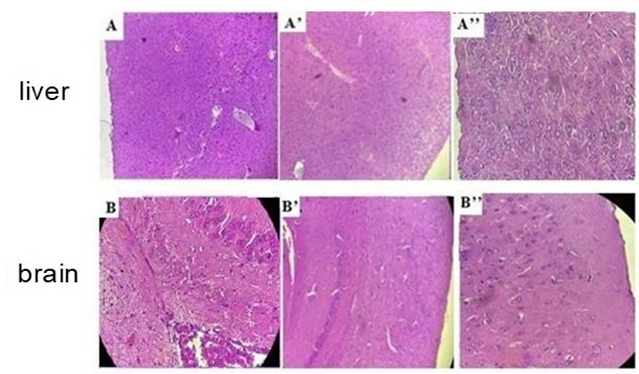
Figure 7. Histological sections of the liver and brain in the group treated with immunostimulants: A, B) Control (×10); A’, B’) Treated (×10); A’’, B’’) Treated (×40). Optical Microscope Leica (×10) and (×40).
Oxidative Stress Parameters
- Catalase (CAT): Graviola induces a significant decrease in hepatic CAT activity (-29.26%) without affecting brain activity. Immunostimulants showed no effect on this enzyme (Figure 8.A), (Figure 8.B).
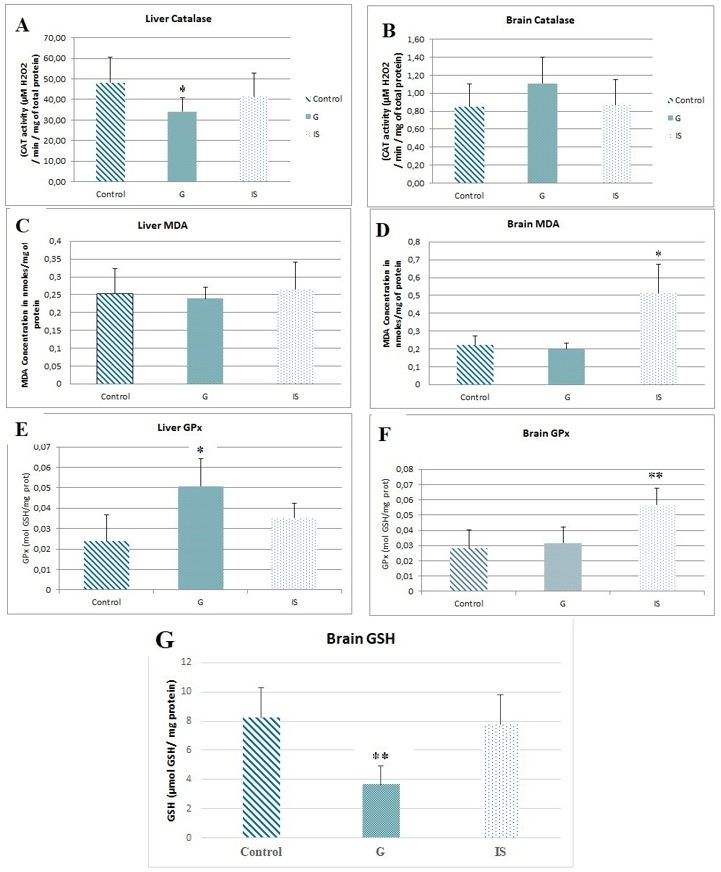
Figure 8. Variations in oxidative stress enzyme activities in mice treated with Graviola (G) or immunostimulants (IS). A) Hepatic catalase activity. B) Brain catalase activity. C) Hepatic glutathione peroxidase (GPx) activity. D) Brain glutathione peroxidase (GPx) activity (*p ≤0.05 vs control; **p≤0.01 vs control). E) Hepatic MDA concentration. F) Brain MDA concentration. G) Brain GSH concentration (*p≤0.05 vs control).
- Malondialdehyde (MDA): Graviola does not affect MDA levels, whereas immunostimulants lead to a significant increase in the brain (+56.68%), with no changes in the liver (Figure 8.C), (Figure 8.D).
- Glutathione Peroxidase (GPx): Hepatic GPx activity significantly increases under Graviola treatment (+53.18%), while immunostimulants cause a significant rise in brain GPx activity (+50.34%) (Figure 8.E), (Figure 8.F).
- Reduced Glutathione (GSH): A marked reduction in brain GSH levels (-55.36%) is observed under Graviola treatment, suggesting a potential neurotoxic effect. Immunostimulants do not influence this parameter (Figure 8.G). Given that the literature reports low catalase activity and high GSH levels in the nervous system, we chose to quantify GSH to assess stress, particularly in relation to the suspected neurodegenerative effects of the plant.
Reproductive Toxicity
Graviola administration reduced the gestation rate in mice, with only 33.33% of females becoming pregnant compared to 100% in the control group. Additionally, a decrease in the average number of offspring was observed (6 compared to 7.83 in the control group). The newborns were counted, and the results are presented in Table 1.
Table 1. Birth outcomes and percentage of pups.
|
Control Group (CG) |
Group G |
Group IS |
% G/CG |
% IS/CG |
% Reduction Compared to Group control |
| G |
IS |
| Pregnant females (Pf) |
6 |
2 |
6 |
33.33 |
100 |
66.66 |
0 |
| Number of pups (Np) |
47 |
12 |
44 |
25.53 |
93.61 |
74.46 |
6.39 |
| Male pups |
23 |
9 |
21 |
39.13 |
91.30 |
60.86 |
8.7 |
| Female pups |
24 |
2 |
23 |
8.33 |
95.83 |
91.66 |
4.17 |
| Np/Pf |
7.83 |
6 |
7.33 |
|
|
|
|
Unlike Graviola, immunostimulants had no impact on reproduction. All treated females became pregnant, and the number of offspring remained similar to that of the control group.
Discussion
The study assessed the subchronic toxicity of Graviola leaf powder and immunostimulants (β-glucan and arabinogalactan) in 18 male Swiss albino mice. Biochemical parameters, oxidative stress, and blood count were analyzed after weekly weight monitoring and animal sacrifice. No significant changes in feeding behavior, systemic disease symptoms, or histopathological abnormalities were observed, suggesting that the tested substances are not toxic.
Graviola
The extract of Annona muricata (Graviola) contains alkaloids and flavonoids (anthocyanins and leuco-anthocyanins), which are known for their biological activities, including anti-inflammatory, antioxidant, and hepatoprotective effects [11,12]. Graviola is traditionally used in tropical regions to treat various ailments. Clinical studies support its hypoglycemic, antioxidant, antimicrobial, and hypotensive properties, but also highlight potential toxicological effects, such as neurotoxicity, severe nausea and vomiting, as well as acute and chronic nephrotoxicity [13-16].
The detection of alkaloids and flavonoids (anthocyanins and leuco-anthocyanins) in the Annona muricata extract suggests that they may induce various biological activities, since flavonoids, a group of phenolic compounds, are involved in several biological effects, such as anti-inflammatory action, antioxidant capacity, and hepatoprotective effects [11,12].
Biochemical Parameters and Complete Blood Count (CBC)
In our study, we observed a 5.1% weight loss in mice treated with the crude Graviola extract (subchronic group), which aligns with the findings of [17], who reported significant weight loss in mice treated for 14 days, despite no reduction in caloric intake [18]. Furthermore, an increase in adiponectin levels was observed in the group treated with 50 mg/kg, suggesting anti-diabetic, anti-inflammatory, and anti-atherogenic effects [19]. High adiponectin levels are associated with reduced risk of type 2 diabetes and improved insulin sensitivity [20].
Therefore, the observed weight loss may be explained by a hypolipidemic effect of the plant, as evidenced by reduced cholesterol and triglyceride levels. On the other hand, the present work showed a slight, non-significant increase in blood glucose levels (+7.08%), consistent with the results of [18], who observed a similar increase in a chronic toxicity study using Graviola extract in male rats.
Our results indicated a highly significant reduction in blood triglyceride levels (-49.03%) in mice treated with Graviola compared to controls. We also noted a slight, non-significant decrease in total cholesterol levels (-24.15%) in treated mice. These findings are consistent with those of Adeyemi et al. (2008), who reported a significant antihyperlipidemic activity of methanolic Graviola extract (100 mg/kg) after two weeks of treatment in streptozotocin-induced diabetic Wistar rats.
According to [21], flavonoids present in Graviola extracts play a major role in the hypolipidemic effect, and also protect LDL (low-density lipoproteins) from oxidative damage, which may help prevent cardiovascular diseases, such as hypertension and atherosclerosis. Additionally, [22] suggest that the presence of flavonoids, tannins, saponins, phytosterols, and phenolic compounds contributes to the hypoglycemic and hypolipidemic effects observed.
The present study demonstrated no significant changes in transaminase levels (AST and ALT) in mice treated with Graviola leaf powder, suggesting no hepatotoxicity at the tested dose, consistent with the findings of [23]. In contrast, [24] reported hepatic enzyme alterations (↑ALT, ↓AST) and dose-dependent signs of toxicity in Wistar rats treated with increasing doses of ethanolic Graviola leaf extract.
In this subchronic toxicity study, Graviola leaf extract did not cause any significant changes in general blood parameters (white blood cells [WBCs], lymphocytes, percentage of lymphocytes, and red blood cells). However, a significant increase in red blood cells (+53.38%) and monocytes (+40.47%) was observed. These findings are consistent with those of [25], who reported a non-significant increase in both red and white blood cell counts in rats treated with 200 mg/kg of dry Graviola extract for eight weeks. In contrast, [26] reported a significant increase (P<0.05) in red blood cells after administering various extracts from different parts of the Graviola plant. This effect was attributed to the presence of alkaloids, flavonoids, and phenolic compounds, which may act on the bone marrow to stimulate or regulate blood cell production.
Oxidative Stress Parameters
In the present study, we observed a significant decrease in hepatic catalase activity, but no change in the brain. This inhibition may be related to a high concentration of superoxide anions. As compensation, hepatic GPx activity increased, while it remained stable in the brain.
The levels of MDA, a marker of lipid peroxidation, did not change, suggesting the absence of oxidative damage to cellular membranes in the liver or brain, which contrasts with the findings of [27].
On the other hand, we observed a marked decrease in brain reduced glutathione (GSH), indicating a high level of oxidative stress. This depletion of GSH is linked to neurological disorders such as Parkinson’s disease, Alzheimer’s, or stroke. These findings are consistent with those of Grover et al., who also reported a decrease in cerebral GSH and genotoxicity associated with the accumulation of reactive oxygen species (ROS).
Histological Analysis
Histological images revealed no tissue abnormalities in the liver or brain, which supports our hematological, biochemical, and oxidative stress results. These findings are consistent with those of [28], who reported no lesions in the liver or pancreas of rats treated with A. stenophylla extract. However, our brain results contradict some epidemiological studies suggesting a link between Graviola consumption and Parkinson's disease. This discrepancy may be explained by the three-month experimental duration.
In contrast, [24] observed liver lesions at doses of 100 and 200 mg/kg, while [29] found that Graviola powder protected against liver damage induced by paracetamol. The protective effects seem to be due to the restoration of redox balance in the liver.
The Acetogenins in Graviola, such as Annonacin, can cross the blood-brain barrier, but their concentration in the brain is limited [30]. Studies suggest that Annonacin may induce an atypical Parkinsonian state due to its neurotoxic effects, resulting from its deposition in nerve cells [16,31,32]. In our study, an immunohistochemical analysis could have detected neurofibrillary tangles, a characteristic feature of neurodegenerative diseases.
Finally, [33] suggested that Graviola leaf extracts could modulate DMN toxicity in the liver due to the presence of chemical compounds like Acetogenins, tannins, phenols, and flavonoids, which enhance endogenous enzyme and antioxidant capacities.
Reproductive Toxicity
The reproduction-based experiment revealed a significant decrease in the litter rate in mice treated with Graviola compared to the control group. Additionally, there was a reduction in the average number of offspring per litter (expressed as the Ns/Fp ratio) in the treated group, suggesting a potential impairment of fertility in male, female, or both mice. No cases of abortion were observed, which aligns with the findings of Damasceno et al. [34], who reported no abortifacient or anti-implantation effects in pregnant rats treated orally with high doses of Annona squamosa aqueous seed extract during the first five days of gestation.
Histological sections of the uterus from Graviola-treated mice showed no architectural abnormalities compared with controls, despite the clear reduction in fertility. Similarly, Damasceno et al. [34] observed no morphological alterations in the uterine endometrium of pregnant rats treated with high doses of Annona squamosa aqueous seed extract during early gestation. Although this study used a different Annona species and extract type, the absence of uterine pathology provides a useful comparison for discussing reproductive effects in our Graviola (Annona muricata) model.
The scientific literature suggests that Graviola may have an effect on:
- Testicles: Affecting sperm production and quality [35,36]. However, [37] contradicted this hypothesis, showing that the ethanolic leaf extract of Graviola might have a positive impact on sperm quality and hormone levels in subjects with testicular dysfunction related to arsenic trioxide.
- Ovaries: Disrupting gamete maturation by affecting hormone secretion [38].
- Hypothalamus and pituitary: Disrupting the regulation of hormones in the hypothalamo-pituitary-gonadal (HPG) axis, including testosterone, luteinizing hormone, and follicle-stimulating hormone. In females, this could lead to disturbances in folliculogenesis and oogenesis, resulting in ovulation failure and uterine cycle issues [39].
- Fallopian tubes: Causing obstruction or dysfunction [40].
All of these hypotheses require further investigation to determine the exact mechanisms by which Graviola affects fertility.
Furthermore, it should be noted that the number of mice used in our experiment is relatively small to definitively confirm the reproductive toxicity of Graviola. We believe that more advanced studies involving a larger sample size of mice will likely provide a more precise answer to this question.
Immunostimulants
The toxicity of immunostimulant polysaccharides, particularly β-glucans and arabinogalactan, has been extensively studied. β-glucans are known for their low toxicity, which supports their potential for therapeutic applications [41,42]. Similarly, arabinogalactan is considered safe by the FDA and is well-tolerated in both humans and animals [43,44].
The findings indicate that β-glucan contributes to lowering lipid levels in the organism. Bae et al. [45] reported that both enzymatically produced β-glucan hydrolysates and native β-glucan led to a marked reduction in serum LDL and VLDL cholesterol, along with an improvement in hepatic lipid parameters in mice. Notably, the hydrolysates promoted a greater fecal elimination of cholesterol and triglycerides than the native form, suggesting a superior lipid-lowering potential. Earlier, Bae et al. [46] reported that oat β-glucan hydrolysates were more effective than native β-glucan in increasing fecal cholesterol and triglyceride excretion in rats fed a high-cholesterol diet.
Biochemical parameters and Complete Blood Count (CBC)
Regarding hematological effects, our study indicated a significant increase in white blood cells (WBCs) and lymphocytes (Ly), which is consistent with previous studies that reported slight but non-significant increases in WBC, Ly, and %Ly, as well as non-significant changes in monocytes and red blood cells (RBC or GR) [47,48]. Clinical studies on arabinogalactan also reported a significant rise in lymphocyte levels [49,50]. These findings suggest a potential synergistic immunostimulatory effect between the two polysaccharides.
On the metabolic level, a slight, non-significant decrease in body weight and blood glucose was observed in the mice treated with immunostimulants, consistent with the results of [51,52]. However, a study by [53] reported a significant reduction in blood glucose following 28 days of oral administration of β-(1→6)-D-glucan from Lasiodiplodia theobromae in mice.
These findings confirm the safety and immunomodulatory potential of the natural immunostimulants used, while highlighting the need for further studies to better understand their full effects.
The consumption of dietary fiber, particularly β-glucans derived from cereals, is well known for its cholesterol-lowering effect [54]. In our study, the administration of an immunostimulant blend led to a significant reduction in blood cholesterol levels, with no change in triglyceride levels. These findings are consistent with those of [55,56], who reported similar effects using yeast and barley-derived β-glucans. On the other hand, [49] showed that arabinogalactan had no impact on lipid parameters, suggesting that the observed hypocholesterolemic effect in the present study is mainly attributable to β-glucan. Viscous β-glucans modulate host cholesterol by affecting bile acid metabolism—they bind to bile acids and prevent their reabsorption in the ileum, increasing their fecal excretion. This, in turn, stimulates the synthesis of new bile acids from cholesterol, thereby lowering systemic LDL cholesterol levels [57].
Regarding transaminases, no significant change was observed in hepatic AST (TGO) levels, while a significant decrease in ALT (TGP) levels was recorded. These findings are partially consistent with [58], who found no significant changes in AST or ALT across various doses of yeast β-glucans. Other studies on β-glucans from different sources support this observation, also reporting no significant changes in these liver enzymes [48,56,59]. Given the limited number of studies addressing arabinogalactan toxicity and its well-established safety profile, it is plausible that the observed decrease in ALT levels is mainly due to β-glucan, suggesting a potential hepatoprotective effect of the immunostimulant mixture.
Oxidative Stress Parameters
β-glucan has been proposed as a regulator of antioxidant enzyme activity (e.g., SOD) and an inhibitor of lipid peroxidation in various animal studies involving rats and rabbits [60,61]. Additionally, arabinogalactan exhibits strong antioxidant activity and is emerging as a safe plant-based source of natural antioxidants with promising biomedical applications [62].
In our study, no significant changes were observed in hepatic catalase or GPx activity, nor in MDA levels, aligning with [63], who reported no changes in antioxidant enzyme activity in rabbits fed a diet containing Pleurotus ostreatus mushrooms. However, in the brain, we observed a highly significant increase in GPx activity and a significant rise in MDA levels, while catalase activity and GSH levels remained unchanged. Similar results were reported by [64], who found increased CAT activity in erythrocytes and enhanced SOD activity in the liver after β-glucan administration. Likewise, Zhang et al. [62] demonstrated that arabinogalactan significantly reduced MDA levels and increased SOD activity in H₂O₂-challenged IEC-6 cells in vitro, indicating antioxidant effects of the polysaccharide.
These findings suggest that the immunostimulant mixture is not systemically toxic, as the liver—its primary detoxifying organ—showed no adverse effects. However, the brain response appears more complex: while elevated GPx activity may reflect a protective response to neutralize free radicals, the concurrent rise in MDA levels suggests that oxidative stress may exceed antioxidant defenses, supporting the hypothesis of a potential pro-oxidant effect in the brain.
Additionally, [65] observed that β-glucan-induced immune stimulation increased antioxidant SOD activity in shrimp (Litopenaeus vannamei).
Histological Analysis
Our histological examination revealed no tissue damage or abnormalities in the livers or brains of mice treated with the immunostimulant mixture compared with the control group. These results are consistent with [47], who, in a study on barley-derived β-glucan, reported no treatment-related lesions in examined organs. Several other studies also highlight the protective effect of β-glucans. For instance, [66] described significant hepatic damage caused by cisplatin, such as sinusoidal congestion, hydropic degeneration, hepatocyte disorganization, and mononuclear cell infiltration. However, when β-glucan was co-administered with cisplatin, these histological alterations were substantially alleviated.
Reproductive Toxicity
Our findings indicated no differences between the control and treated groups concerning reproductive toxicity, supporting the safety of the immunostimulant mixture—even at doses ten times higher than standard—regarding fertility or general toxic effects.
Conclusions
This study assessed the subchronic effects of Graviola leaf powder and a mixture of natural immunostimulants (β-glucan and arabinogalactan) in mice. Both treatments showed no signs of general toxicity, with normal histological architecture in major organs and no significant changes in key biochemical parameters.
Graviola exhibited hypolipidemic and hematological effects, but also showed potential neurotoxicity (GSH depletion in the brain) and reproductive toxicity, marked by reduced fertility. These effects suggest the need for caution and further investigation into its safety profile.
In contrast, the immunostimulants demonstrated favorable immunomodulatory and metabolic effects without adverse impacts on the liver, brain, or reproductive system, supporting their good safety and therapeutic potential.
Further studies are recommended to clarify long-term effects and underlying mechanisms, especially regarding Graviola’s possible neuro- and reproductive toxicity.
Note: This study was derived from the PhD thesis of Mohammed Abdellaoui, completed at Université Badji Mokhtar, Annaba, Algeria.
Data Availability
All data generated from the mouse experiments and analyzed using the Microsoft Excel software are available from the corresponding author upon reasonable request.
Ethical Considerations
This work involved animal experiments conducted in accordance with ethical guidelines for animal research, following Directive 2010/63/EU on the protection of animals used for scientific purposes and the regulations of the AASAE (Algerian Association for Animal Experimentation Sciences), under Authorization Number 45/DGLPAG/DVA.SDA.14.
Authors' Contributions
MA: Conceptualization; Methodology; Formal analysis; Investigation; Writing – Original Draft
AK: Conceptualization; Methodology; Validation; Supervision; Writing – Review & Editing –
FZM: Data Curation; Formal analysis
YK: Investigation; Visualization
MM: Resources; Project administration
Acknowledgement
None.
Conflict of Interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
This study was supported by the personal financial contribution of the first author and the facilities provided by the University of Badji Mokhtar, Annaba, Algeria.
References
- Saripalli HR, Dixit PK. In vitro studies on anti-tumour activities of solvent extracts of leaves, fruit pulp and seeds of graviola (Gishta) cultivar of Ethiopia, N. E. Africa. Int J Innov Res Sci Eng Technol. 2017;6(8):16743-9. [DOI: 10.15680/IJIRSET.2017.0608001]
- Daddiouaissa D, Amid A, Kabbashi NA, Fuad FAA, Elnour AM, Epandy MAKMS. Antiproliferative activity of ionic liquid-graviola fruit extract against human breast cancer (MCF-7) cell lines using flow cytometry techniques. J Ethnopharmacol. 2019;236:466-73. [DOI: 10.1016/j.jep.2019.03.003] [PMID: 30853648]
- Daddiouaissa D, Amid A, Shirwan MA, Elnour AM. Phytochemical analysis of ionic liquid-Graviola (Annona muricata) fruit extract and its acute toxicity on zebrafish early-life stages. Asia Pac J Mol Biol Biotechnol. 2020;28(2):113-24. [DOI: 10.35118/apjmbb.2020.028.2.10]
- Caparros-Lefebvre D, Elbaz A. Possible relation of atypical parkinsonism in the French West Indies with consumption of tropical plants: a case-control study. Caribbean Parkinsonism Study Group. Lancet. 1999;354(9175):281-6. [DOI: 10.1016/s0140-6736(98)10166-6] [PMID: 10440304]
- Rottscholl R, Haegele M, Jainsch B, Xu H, Respondek G, Höllerhage M, et al. Chronic consumption of Annona muricata juice triggers and aggravates cerebral tau phosphorylation in wild-type and MAPT transgenic mice. J Neurochem. 2016;139(4):624-39. [DOI: 10.1111/jnc.13835] [PMID: 27569447]
- Smith RE, Shejwalkar P. Potential neurotoxicity of graviola (Annona muricata) juice. InSafety issues in beverage production. 2020;18:429-49. [DOI: 10.1016/B978-0-12-816679-6.00013-9]
- Hong F, Yan J, Baran JT, Allendorf DJ, Hansen RD, Ostroff GR, et al. Mechanism by which orally administered beta-1,3-glucans enhance the tumoricidal activity of antitumor monoclonal antibodies in murine tumor models. J Immunol. 2004;173(2):797-806. [DOI: 10.4049/jimmunol.173.2.797] [PMID: 15240666]
- Li B, Allendorf DJ, Hansen R, Marroquin J, Ding C, Cramer DE, et al. Yeast beta-glucan amplifies phagocyte killing of iC3b-opsonized tumor cells via complement receptor 3-Syk-phosphatidylinositol 3-kinase pathway. J Immunol. 2006;177(3):1661-9. [DOI: 10.4049/jimmunol.177.3.1661] [PMID: 16849475]
- Kelly GS. Larch arabinogalactan: clinical relevance of a novel immune-enhancing polysaccharide. Altern Med Rev. 1999;4(2):96-103. [PMID: 10231609]
- Notions - Comment évaluer un effet toxique ? - Cnesst. 2024. [LINK]
- Ortuño A, Báidez A, Gómez P, Arcas MC, Porras I, García-Lidón A, et al. Citrus paradisi and Citrus sinensis flavonoids: Their influence in the defence mechanism against Penicillium digitatum. Food Chem. 2006;98(2):351-8. [DOI: 10.1016/j.foodchem.2005.06.017]
- Arnaud K, Nicodème C, Durand DN, Martial N, Basile S, Haziz S, et al. Antioxidant, anti-inflammatory efficacy and HPLC analysis of Annona muricata leaves extracts from Republic of Benin. Am J Plant Sci. 2020;11(6):803-18. [DOI: 10.4236/ajps.2020.116057]
- Arroyo J, Martínez J, Ronceros G, Palomino R, Villarreal A, Bonilla P, et al. Efecto hipoglicemiante coadyuvante del extracto etanólico de hojas de Annona muricata L (guanábana), en pacientes con diabetes tipo 2 bajo tratamiento de glibenclamida. An Fac Med. 2009;70(3):163-7. [LINK]
- Coria-Téllez AV, Montalvo-Gónzalez E, Yahia EM, Obledo-Vázquez EN. Annona muricata: A comprehensive review on its traditional medicinal uses, phytochemicals, pharmacological activities, mechanisms of action and toxicity. Arab J chem. 2018;11(5):662-91. [DOI: 10.1016/j.arabjc.2016.01.004]
- Coria-Téllez AV, Montalvo-Gónzalez E, Yahia EM, Obledo-Vázquez EN. Annona muricata: a comprehensive review on its traditional medicinal uses, phytochemicals, pharmacological activities, mechanisms of action and toxicity. Arab J Chem. 2018;11(5):662-91. [DOI: 10.1016/j.arabjc.2016.01.004]
- Ferreira GG, Quaresma ACS, Brandão DLDN, Marinho AMDR, Siqueira JES, Correa KL, et al. Evaluation of genotoxicity and toxicity of Annona muricata L. Seeds and in silico studies. Mol. 2022;28(1):231. [DOI: 10.3390/molecules28010231] [PMID: 36615425]
- Sasso S, Sampaio E Souza PC, Santana LF, Cardoso CAL, Alves FM, Portugal LC, et al. Use of an extract of Annona muricata linn to prevent high-fat diet induced metabolic disorders in C57BL/6 Mice. Nutr. 2019;11(7):1509. [DOI: 10.3390/nu11071509] [PMID: 31269728]
- Radapong S, Suppajariyawat P, Sincharoenpokai P, Chaorai B, Phankhajon K, Primprai P, et al. Chronic toxicity of Annona muricata L. leaf extract. Agriculture Nat Resources. 2019;53(1):53-60. [LINK]
- Scoditti E, Massaro M, Carluccio MA, Pellegrino M, Wabitsch M, Calabriso N, et al. Additive regulation of adiponectin expression by the mediterranean diet olive oil components oleic acid and hydroxytyrosol in human adipocytes. Plos One. 2015;10(6):e0128218. [DOI: 10.1371/journal.pone.0128218] [PMID: 26030149]
- Begum M, Choubey M, Tirumalasetty MB, Arbee S, Mohib MM, Wahiduzzaman M, et al. Adiponectin: A promising target for the treatment of diabetes and its complications. Life (Basel). 2023;13(11):2213. [DOI: 10.3390/life13112213] [PMID: 38004353]
- Sandhar HK, Kumar B, Prasher S, Tiwari P, Salhan M, Sharma P. A review of phytochemistry and pharmacology of flavonoids. Int pharm sci. 2011;1(1):25-41. [LINK]
- Sovia E, Ratwita W, Wijayanti D, Novianty DR. Hypoglycemic and hypolipidemic effects of Annona muricata L. leaf ethanol extract. Int J Pharm Pharm Sci. 2017;9(3):170-4. [DOI: 10.22159/ijpps.2017v9i3.16402]
- Zubaidi SN, Qadi WSM, Maarof S, Mohmad Misnan N, Mohammad Noor HS, et al. Assessing the acute toxicological effects of Annona muricata leaf ethanol extract on rats: biochemical, histopathological, and metabolomics analyses. Toxics. 2023;11(8):688. [DOI: 10.3390/toxics11080688] [PMID: 37624193]
- Natacha A, Melanie A, Behanzin J, Alphonse S. Evaluation of the toxicity of Annona muricata leaf extracts on liver and kidney function and investigation of acute and subacute toxicity in Wistar rats. Am J Pharm Tech Res. 2018;8(1). [LINK]
- Shukry M, El-Shehawi AM, El-Kholy WM, Elsisy RA, Hamoda HS, Tohamy HG, et al. Ameliorative effect of Graviola (Annona muricata) on mono sodium glutamate-induced hepatic injury in rats: antioxidant, apoptotic, anti-inflammatory, lipogenesis markers, and histopathological studies. Animals (Basel). 2020;10(11):1996. [DOI: 10.3390/ani10111996] [PMID: 33143024]
- Agu KC, Okolie NP, Eze I, Anionye JC, Falodun A. Phytochemical analysis, toxicity profile, and hemomodulatory properties of Annona muricata (Soursop). Egypt J Haematol. 2017;42(1):36-44. [DOI: 10.4103/1110-1067.206431]
- Grover P, Singh SP, Prabhakar PV, Reddy UA, Balasubramanyam A, Mahboob M, et al. In vivo assessment of genotoxic effects of Annona squamosa seed extract in rats. Food Chem Toxicol. 2009;47(8):1964-71. [DOI: 10.1016/j.fct.2009.05.013] [PMID: 19457440]
- Chagonda LS, Tafadzwa M, Dexter T, Louis G, Exnevia G, Felicity B, et al. Acute and sub-acute oral toxicity of hydroethanolic root extract of annona stenophylla engl. and diels in sprague dawley rats. J Biol Active Product Nat. 2015;5(5):349-56. [DOI: 10.1080/22311866.2015.1110501]
- Menon S, Al-Eisa RA, Hamdi H, Lawrence L, Syamily PS, Sivaram VP, et al. Protective effect of Annona muricata Linn fruit pulp lyophilized powder against paracetamol-induced redox imbalance and hepatotoxicity in rats. Processes. 2023;11(1):276. [DOI: 10.3390/pr11010276]
- Bonneau N, Schmitz-Afonso I, Brunelle A, Touboul D, Champy P. Method development for quantification of the environmental neurotoxin annonacin in rat plasma by UPLC-MS/MS and application to a pharmacokinetic study. J Chromatogr B Analyt Technol Biomed Life Sci. 2015;1004:46-52. [DOI: 10.1016/j.jchromb.2015.09.039] [PMID: 26444335]
- Champy P, Höglinger GU, Féger J, Gleye C, Hocquemiller R, Laurens A, et al. Annonacin, a lipophilic inhibitor of mitochondrial complex I, induces nigral and striatal neurodegeneration in rats: possible relevance for atypical parkinsonism in Guadeloupe. J Neurochem. 2004;88(1):63-9. [DOI: 10.1046/j.1471-4159.2003.02138.x] [PMID: 14675150]
- Escobar-Khondiker M, Höllerhage M, Muriel MP, Champy P, Bach A, Depienne C, et al. Annonacin, a natural mitochondrial complex I inhibitor, causes tau pathology in cultured neurons. J Neurosci. 2007;27(29):7827-37. [DOI: 10.1523/JNEUROSCI.1644-07.2007] [PMID: 17634376]
- Usunobun U. Protective effects of Annona muricata ethanolic leaf extract against dimethylnitrosamine (DMN)-induced hepatotoxicity. IOSR J Pharm Biol Sci. 2014;9(4):1-6. [DOI: 10.9790/3008-09420106]
- Damasceno DC, Volpato GT, Sartori TC, Rodrigues PF, Perin EA, Calderon IM, et al. Effects of Annona squamosa extract on early pregnancy in rats. Phytomed. 2002;9(7):667-72. [DOI: 10.1078/094471102321616508] [PMID: 12487333]
- Borg CL, Wolski KM, Gibbs GM, O'Bryan MK. Phenotyping male infertility in the mouse: how to get the most out of a 'non-performer'. Hum Reprod Update. 2010;16(2):205-24. [DOI: 10.1093/humupd/dmp032] [PMID: 19758979]
- Hou B, Wang F, Liu T, Wang Z. Reproductive toxicity of polystyrene microplastics: In vivo experimental study on testicular toxicity in mice. J Hazard Mater. 2021;405:124028. [DOI: 10.1016/j.jhazmat.2020.124028] [PMID: 33087287]
- Eyeghre OA, Dike CC, Ezeokafor EN, Oparaji KC, Amadi CS, Chukwuma CC, et al. The impact of Annona muricata and metformin on semen quality and hormonal profile in Arsenic trioxide-induced testicular dysfunction in male Wistar rats. Magna Sci Adv Res Rev. 2023;8(01):001-18. [DOI: 10.30574/msarr.2023.8.1.0053]
- Pelusi C, Ikeda Y, Zubair M, Parker KL. Impaired follicle development and infertility in female mice lacking steroidogenic factor 1 in ovarian granulosa cells. Biol Reprod. 2008;79(6):1074-83. [DOI: 10.1095/biolreprod.108.069435] [PMID: 18703422]
- Piomboni P, Governini L, Gori M, Puggioni E, Costantino-Ceccarini E, Luddi A. New players in the infertility of a mouse model of lysosomal storage disease: the hypothalamus-pituitary-gonadal axis. Front Endocrinol (Lausanne). 2014;4:204. [DOI: 10.3389/fendo.2013.00204] [PMID: 24432014]
- Shen H, Cai M, Chen T, Zheng D, Huang S, Zhou M, et al. Factors affecting the success of fallopian tube recanalization in treatment of tubal obstructive infertility. J Int Med Res. 2020;48(12):300060520979218. [DOI: 10.1177/0300060520979218] [PMID: 33322979]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the safety of ‘yeast beta-glucans’ as a Novel Food ingredient. EFSAJ. 2011;9(5). [DOI: 10.2903/j.efsa.2011.2137]
- Barton C, Vigor K, Scott R, Jones P, Lentfer H, Bax HJ, et al. Beta-glucan contamination of pharmaceutical products: How much should we accept? Cancer Immunol Immunother. 2016;65(11):1289-1301. [DOI: 10.1007/s00262-016-1875-9] [PMID: 27473075]
- Groman EV, Enriquez PM, Jung C, Josephson L. Arabinogalactan for hepatic drug delivery. Bioconjug Chem. 1994;5(6):547-56. [DOI: 10.1021/bc00030a010] [PMID: 7533005]
- Ash ME. A review of larch arabinogalactans. Clin Edu. 2016. [LINK]
- Bae IY, Lee S, Kim SM, Lee HG. Effect of partially hydrolyzed oat β-glucan on the weight gain and lipid profile of mice. Food Hydrocolloids. 2009;23(7):2016-21. [DOI: 10.1016/j.foodhyd.2009.03.016]
- Bae IY, Kim SM, Lee S, Lee HG. Effect of enzymatic hydrolysis on cholesterol-lowering activity of oat beta-glucan. N Biotechnol. 2010;27(1):85-8. [DOI: 10.1016/j.nbt.2009.11.003] [PMID: 19931657]
- Delaney B, Carlson T, Frazer S, Zheng T, Hess R, Ostergren K, et al. Evaluation of the toxicity of concentrated barley beta-glucan in a 28-day feeding study in Wistar rats. Food Chem Toxicol. 2003;41(4):477-87. [DOI: 10.1016/s0278-6915(02)00298-3] [PMID: 12615121]
- Chen SN, Nan FH, Chen S, Wu JF, Lu CL, Soni MG. Safety assessment of mushroom β-glucan: subchronic toxicity in rodents and mutagenicity studies. Food Chem Toxicol. 2011;49(11):2890-8. [DOI: 10.1016/j.fct.2011.08.007] [PMID: 21856366]
- Kim LS, Burkholder PM, Waters RF. Effects of low-dose larch arabinogalactan from larix occidentalis: a randomized, double-blind, placebo-controlled pilot study. Complement Health Pract Rev. 2002;7(3):221-9. [DOI: 10.1177/153321010200700305]
- Dion C, Chappuis E, Ripoll C. Does larch arabinogalactan enhance immune function? A review of mechanistic and clinical trials. Nutr Metab (Lond). 2016;13:28. [DOI: 10.1186/s12986-016-0086-x] [PMID: 27073407]
- Williams DL, Sherwood ER, Browder IW, McNamee RB, Jones EL, Di Luzio NR. Pre-clinical safety evaluation of soluble glucan. Int J Immunopharmacol. 1988;10(4):405-14. [DOI: 10.1016/0192-0561(88)90127-0] [PMID: 3262594]
- Canaan MM, Reis-Canaan JC, Zangerônimo MG, Andrade EF, Gonçalves TMSV, Pereira MCA, et al. Yeast Beta-glucans ingestion does not influence body weight: a systematic review and meta-analysis of pre-clinical studies. Nutr. 2021;13(12):4250. [DOI: 10.3390/nu13124250] [PMID: 34959802]
- Túrmina JA, Carraro E, Alves da Cunha MA, Dekker RF, Barbosa AM, Dos Santos FS, et al. Toxicological assessment of β-(1-->6)-glucan (lasiodiplodan) in mice during a 28-day feeding study by gavage. Mol. 2012;17(12):14298-309. [DOI: 10.3390/molecules171214298] [PMID: 23208465]
- Brown L, Rosner B, Willett WW, Sacks FM. Cholesterol-lowering effects of dietary fiber: a meta-analysis. Am J Clin Nutr. 1999;69(1):30-42. [DOI: 10.1093/ajcn/69.1.30] [PMID: 9925120]
- Nicolosi R, Bell SJ, Bistrian BR, Greenberg I, Forse RA, Blackburn GL. Plasma lipid changes after supplementation with beta-glucan fiber from yeast. Am J Clin Nutr. 1999;70(2):208-12. [DOI: 10.1093/ajcn.70.2.208] [PMID: 10426696]
- Jonker D, Hasselwander O, Tervilä-Wilo A, Tenning PP. 28-Day oral toxicity study in rats with high purity barley beta-glucan (Glucagel). Food Chem Toxicol. 2010;48(1):422-8. [DOI: 10.1016/j.fct.2009.10.034] [PMID: 19879311]
- Joyce SA, Kamil A, Fleige L, Gahan CGM. The Cholesterol-lowering effect of oats and oat beta glucan: modes of action and potential role of bile acids and the microbiome. Front Nutr. 2019;6:171. [DOI: 10.3389/fnut.2019.00171] [PMID: 31828074]
- Babícek K, Cechová I, Simon RR, Harwood M, Cox DJ. Toxicological assessment of a particulate yeast (1,3/1,6)-beta-D-glucan in rats. Food Chem Toxicol. 2007;45(9):1719-30. [DOI: 10.1016/j.fct.2007.03.013] [PMID: 17493735]
- Chen SN, Chang CS, Chen S, Soni M. Subchronic toxicity and genotoxicity studies of Antrodia mushroom β-glucan preparation. Regul Toxicol Pharmacol. 2018;92:429-38. [DOI: 10.1016/j.yrtph.2017.12.022] [PMID: 29287801]
- Pietrzycka A, Stepniewski M, Waszkielewicz AM, Marona H, Krzyzanowska A, Kłosowska K, et al. Effect of vita glucan on some antioxidant parameters of the human blood. In vitro study. Acta Pol Pharm. 2006;63(6):547-51. [PMID: 17438873]
- Toklu HZ, Sener G, Jahovic N, Uslu B, Arbak S, Yeğen BC. Beta-glucan protects against burn-induced oxidative organ damage in rats. Int Immunopharmacol. 2006;6(2):156-69. [DOI: 10.1016/j.intimp.2005.07.016] [PMID: 16399620]
- Zhang S, Wang S, Fan YY, Liu WC, Zheng YN, Wang Z, et al. Preparation of a new resource food-arabinogalactan and its protective effect against enterotoxicity in IEC-6 cells by inhibiting endoplasmic reticulum stress. Int J Biol Macromol. 2023;249:126124. [DOI: 10.1016/j.ijbiomac.2023.126124] [PMID: 37543271]
- Bobek P, Galbavý S. Hliva ústricovitá (Pleurotus ostreatus) efektívne bráni rozvoju aterosklerózy u králika [The oyster mushroom (Pleurotus ostreatus) effectively prevents the development of atherosclerosis in rabbits]. Ceska Slov Farm. 1999;48(5):226-30. [PMID: 10566243]
- Bobek P, Galbavy S. Effect of pleuran (beta-glucan from Pleurotus ostreatus) on the antioxidant status of the organism and on dimethylhydrazine-induced precancerous lesions in rat colon. Br J Biomed Sci. 2001;58(3):164-8. [PMID: 11575739]
- Campa-Córdova AI, Hernández-Saavedra NY, Ascencio F. Superoxide dismutase as modulator of immune function in American white shrimp (Litopenaeus vannamei). Comp Biochem Physiol C Toxicol Pharmacol. 2002;133(4):557-65. [DOI: 10.1016/s1532-0456(02)00125-4] [PMID: 12458183]
- Yılmaz BD, Eren B, Sağır D, Başardı A, Mercan S. Histological evaluation of the protective role of β-glucan against cisplatin-induced hepatotoxicity. Karamanoğlu Mehmetbey Üniversitesi Mühendislik ve Doğa Bilimleri Dergisi. 2024;6(1):38-43. [DOI: 10.55213/kmujens.1434724]















































